Ligand-Based vs. Target-Based Chemogenomics: A Strategic Guide for Modern Drug Discovery
This article provides a comprehensive comparison of ligand-based and target-based chemogenomic approaches, two foundational strategies in contemporary drug discovery.
Ligand-Based vs. Target-Based Chemogenomics: A Strategic Guide for Modern Drug Discovery
Abstract
This article provides a comprehensive comparison of ligand-based and target-based chemogenomic approaches, two foundational strategies in contemporary drug discovery. Aimed at researchers, scientists, and drug development professionals, it explores the core principles, methodologies, and practical applications of each paradigm. The content delves into the distinct advantages and limitations of both approaches, offers strategies for troubleshooting and optimization, and presents a framework for their rigorous validation and comparative analysis. By synthesizing insights from current literature and case studies, this guide aims to empower scientists to select and integrate the most effective computational strategies for target identification, lead optimization, and drug repurposing, ultimately enhancing the efficiency and success rate of drug development pipelines.
Core Principles: Unveiling the Foundations of Chemogenomic Strategies
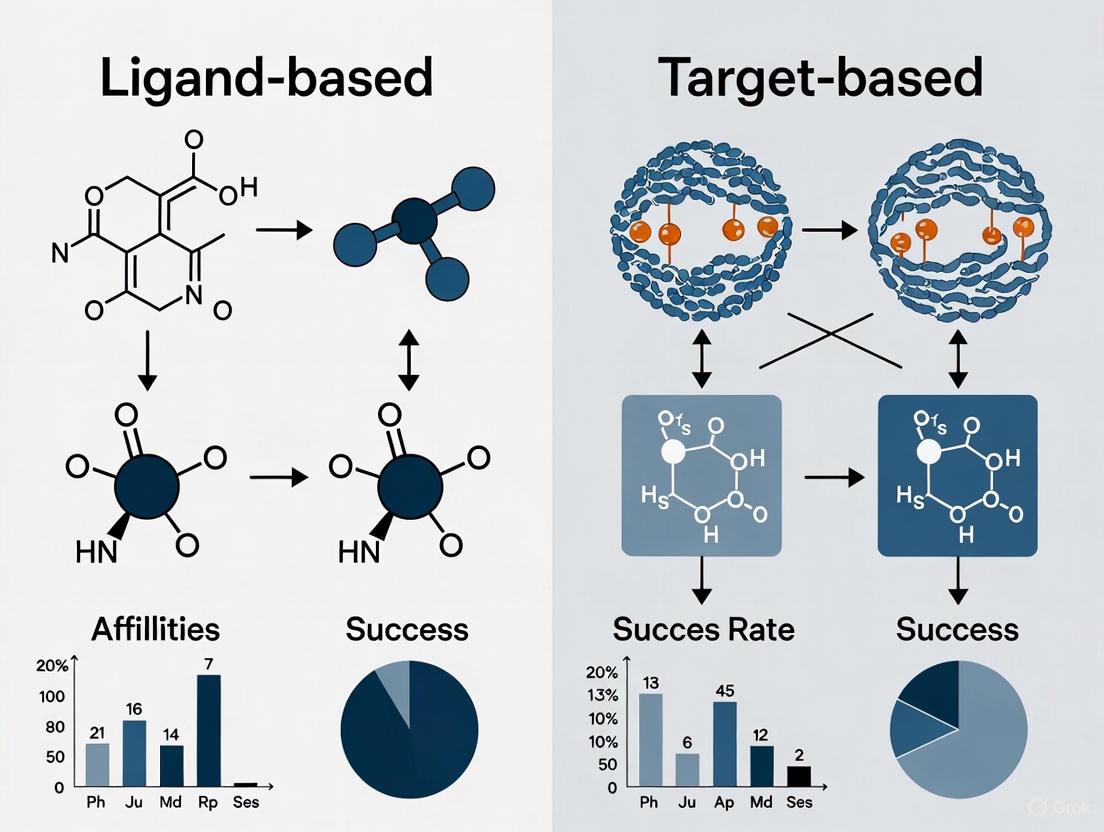
In the field of chemogenomics, which systematically explores the interaction between chemical space and biological target space, two primary computational strategies have emerged for drug discovery and target prediction: ligand-based and target-based approaches [1] [2] [3]. The core premise linking these paradigms is that similar ligands tend to bind to similar targets, and conversely, similar targets tend to bind to similar ligands [1] [3]. This guide provides a comparative analysis of these two methodologies, supported by recent performance data and detailed experimental protocols.
Core Principles and Methodologies
The fundamental difference between these approaches lies in their starting point and the primary information they utilize for drug discovery and target prediction.
Ligand-Based Approaches
Ligand-based methods rely on the principle that compounds with similar chemical structures are likely to have similar biological activities [1] [4]. These approaches do not require explicit structural information about the target protein.
- Key Techniques: The most common techniques include chemical similarity searching using molecular fingerprints, pharmacophore modeling, and Quantitative Structure-Activity Relationship (QSAR) analysis [1] [5]. These methods describe molecules using 1D, 2D, or 3D molecular descriptors [1].
- Similarity Measurement: Structural similarity is typically quantified using the Tanimoto coefficient, which compares molecular fingerprints such as Morgan fingerprints (ECFP-like) or FP2 fingerprints [1] [4]. For example, the MOST (MOst-Similar ligand-based Target inference) approach uses fingerprint similarity and explicit bioactivity data of the most-similar ligands to predict targets with high accuracy [4].
Target-Based Approaches
Target-based methods, in contrast, begin with the protein target of interest. These approaches leverage the three-dimensional structure of the target to identify or design potential ligands.
- Key Techniques: The predominant technique is molecular docking, which predicts how a small molecule binds to a protein's active site and scores the interaction strength [6] [7]. Structure-based virtual screening can systematically dock chemical libraries against arrays of protein targets [8].
- Requirements: These methods depend on the availability of high-quality 3D protein structures, which can come from experimental methods like X-ray crystallography or from computational models generated by tools like AlphaFold [6].
Table 1: Fundamental Characteristics of Ligand-Based and Target-Based Approaches
| Characteristic | Ligand-Based Approaches | Target-Based Approaches |
|---|---|---|
| Starting Point | Known active ligands | Protein target structure |
| Core Principle | "Similar ligands have similar activities" | "Structure-based molecular recognition" |
| Target Info Required | No 3D structure needed | High-quality 3D structure essential |
| Primary Techniques | Chemical similarity searching, QSAR, pharmacophore modeling | Molecular docking, structure-based virtual screening |
| Data Sources | ChEMBL, DrugBank, BindingDB [6] | PDB, AlphaFold models [6] |
Performance Comparison and Experimental Data
Recent systematic comparisons provide quantitative insights into the performance of these approaches. A 2025 benchmark study evaluated seven target prediction methods using a shared dataset of FDA-approved drugs, offering direct performance comparisons [6].
Table 2: Performance Comparison of Representative Target Prediction Methods
| Method Name | Approach Type | Algorithm/Fingerprint | Key Performance Findings |
|---|---|---|---|
| MolTarPred | Ligand-based | 2D similarity, MACCS/Morgan fingerprints | Most effective method in 2025 benchmark; Morgan fingerprints with Tanimoto outperformed MACCS with Dice scores [6] |
| MOST | Ligand-based | Morgan/FP2 fingerprints with machine learning | Achieved 87-95% accuracy in cross-validation; Morgan fingerprint slightly better than FP2 [4] |
| RF-QSAR | Target-centric | Random forest, ECFP4 | Included in systematic comparison [6] |
| TargetNet | Target-centric | Naïve Bayes, multiple fingerprints | Included in systematic comparison [6] |
| CMTNN | Target-centric | Neural network, Morgan fingerprints | Included in systematic comparison [6] |
| D3Similarity | Ligand-based | 2D and 3D similarity combination | Complementary to structure-based docking; uses 2D × 3D value as integrated score [9] |
Performance Trade-offs and Optimization
The 2025 comparison revealed that model optimization strategies involve important trade-offs. For instance, applying high-confidence filtering to interaction data, while improving precision, reduces recall, making it less ideal for drug repurposing applications where maximizing potential leads is prioritized [6].
Experimental Protocols
To illustrate how these approaches are implemented in practice, here are detailed methodologies from key studies.
Protocol 1: Ligand-Based Target Prediction (MOST Approach)
The MOST approach provides a robust protocol for ligand-based target inference [4]:
- Data Preparation: Extract bioactivity data (e.g., Ki values) from curated databases like ChEMBL. Filter for direct binding data with high confidence scores and resolve multiple measurements for the same target-ligand pair.
- Fingerprint Calculation: Generate molecular fingerprints for all compounds. The Morgan fingerprint (radius = 2) is recommended based on its superior performance.
- Similarity Search: For each query compound, calculate Tanimoto coefficients against all ligands in the training database. Identify the most-similar ligand(s) for each target.
- Model Training: Use machine learning methods (Logistic Regression or Random Forest performed best) to learn the relationship between fingerprint similarity, bioactivity of the most-similar ligand, and the probability of the query compound being active.
- Target Prediction: Apply the trained model to predict potential targets for new query compounds, using explicit bioactivity data (pKi values) of the most-similar ligands to calculate statistical significance (p-values) and control false discovery rates in multiple-target predictions.
Protocol 2: Structure-Based Virtual Screening
A standard protocol for target-based screening involves [6] [7]:
- Target Preparation: Obtain the 3D structure of the target protein from PDB or generate it using computational tools like AlphaFold. Prepare the structure by adding hydrogen atoms, assigning partial charges, and optimizing side-chain conformations.
- Binding Site Identification: Define the binding pocket, either from known co-crystallized ligands or through binding site prediction algorithms.
- Ligand Library Preparation: Curate a library of small molecules for screening, ensuring correct protonation states, tautomers, and 3D conformations.
- Molecular Docking: Systematically dock each compound from the library into the binding site using programs like AutoDock Vina or Glide. Generate multiple binding poses per compound.
- Scoring and Ranking: Use scoring functions to evaluate and rank the binding poses. Apply consensus scoring or post-processing with machine-learning-based scoring functions to improve accuracy.
- Experimental Validation: Select top-ranked compounds for in vitro testing to validate the predictions.
Successful implementation of these approaches requires access to specialized databases, software tools, and computational resources.
Table 3: Essential Resources for Chemogenomics Research
| Resource Category | Specific Tools/Databases | Key Function |
|---|---|---|
| Bioactivity Databases | ChEMBL, BindingDB, DrugBank | Provide curated ligand-target interaction data for ligand-based approaches [6] [8] |
| Protein Structure Resources | Protein Data Bank (PDB), AlphaFold Database | Source of 3D protein structures for target-based approaches [6] [8] |
| Cheminformatics Toolkits | RDKit, Open Babel | Calculate molecular fingerprints, perform structural manipulations, and similarity searching [9] [4] |
| Docking Software | AutoDock Vina, Glide, GOLD | Perform molecular docking and structure-based virtual screening [7] |
| Specialized Target Prediction Tools | MolTarPred, MOST, SEA, D3Similarity | Integrated platforms for predicting drug targets using various algorithms [6] [9] [4] |
Application Contexts and Decision Framework
The choice between ligand-based and target-based approaches depends on the specific research question, data availability, and project goals.
When to Prefer Ligand-Based Approaches
- Novel Target Identification: When discovering new protein targets for existing drugs or natural products with known phenotypic effects [2] [5].
- Limited Structural Data: When high-quality 3D structures of the target protein are unavailable [3].
- Drug Repurposing: To identify new therapeutic indications for existing drugs by predicting off-target effects [6] [4].
- Natural Products Research: For elucidating mechanisms of action of traditional medicines or natural products with complex structures [2] [5].
When to Prefer Target-Based Approaches
- Novel Lead Identification: When searching for new chemical starting points against a well-characterized target [7].
- Rational Drug Design: To optimize lead compounds through structure-based design when protein-ligand complex structures are available [7].
- Selectivity Profiling: To design selective compounds for specific protein family members by exploiting differences in binding sites [5].
- Target-Hopping: To identify ligands for orphan receptors with known structures but limited ligand information [3].
Emerging Trends and Integrated Approaches
The distinction between ligand-based and target-based paradigms is increasingly blurring with the emergence of integrated strategies [8] [10]. Modern chemogenomics leverages both chemical and biological information to create more accurate predictive models.
Hybrid Target-Ligand Approaches
These methods simultaneously consider descriptors of both ligands and proteins, creating unified models that can predict interactions across broader sections of the protein-ligand space [8]. For instance, some approaches merge protein sequence descriptors with ligand fingerprints to create interaction models that can generalize to proteins with limited ligand information [8].
Artificial Intelligence and Machine Learning
Recent advances incorporate deep learning and knowledge graphs to significantly improve target prediction accuracy [5]. These systems offer multi-dimensional drug-target interaction analysis, integrate multi-omics data, and provide interpretable decision support with clinical translatability [5].
The field continues to evolve with improvements in both approaches: ligand-based methods are incorporating more sophisticated similarity metrics and explicit bioactivity data [4], while target-based methods benefit from more accurate protein structure prediction and machine-learning scoring functions [6].
Decision Framework for Selecting Between Ligand-Based and Target-Based Approaches
Both ligand-based and target-based approaches offer distinct advantages for drug discovery within the chemogenomics paradigm. Ligand-based methods excel when sufficient ligand bioactivity data exists, providing high prediction accuracy and efficiency for applications like drug repurposing. Target-based approaches offer rational structure-based design capabilities when reliable protein structures are available. The most effective drug discovery strategies often integrate both approaches, leveraging their complementary strengths to navigate the complex landscape of protein-ligand interactions. As both methodologies continue to advance—with improvements in similarity metrics, machine learning scoring functions, and hybrid models—their impact on accelerating drug discovery and understanding polypharmacology will continue to grow.
The accurate prediction of interactions between a drug and its target protein is a cornerstone of modern drug discovery, serving as an efficient analog to costly and time-consuming wet-lab experiments [11]. The foundational principles guiding this field have evolved into two primary computational philosophies: the ligand-based paradigm, which operates on the "guilt-by-association" principle that similar ligands tend to bind similar targets, and the target-based paradigm, which prioritizes structural complementarity between a drug and the three-dimensional (3D) structure of its protein target [12] [10]. Within these paradigms, chemogenomic approaches that systematically study the interaction of small molecules with macromolecular target families have gained prominence [11]. This guide provides an objective comparison of these methodologies, evaluating their performance, detailing experimental protocols, and situating them within the broader context of chemogenomic research for an audience of researchers, scientists, and drug development professionals.
Methodological Foundations and Comparative Workflow
The two approaches, ligand-based and target-based, offer distinct yet complementary paths for predicting drug-target interactions (DTIs). The ligand-based approach relies on the similarity principle, where a query molecule is compared to a database of known active ligands; if the query is highly similar to a known ligand, it is predicted to bind the same target [6] [11]. In contrast, the target-based approach uses the 3D structure of a protein target to perform molecular docking, simulating how a query compound might fit and bind within a specific binding site, thereby prioritizing structural complementarity [13] [10]. A third, hybrid category known as proteochemometric modeling (PCM) has emerged, which integrates information from both multiple ligands and related protein targets into a single machine-learning model, allowing for both inter- and extrapolation to novel compounds and targets [12].
The diagram below illustrates the logical relationships and workflows connecting these core principles to their corresponding methodologies and data requirements.
Performance Comparison of Representative Methods
A precise comparative study published in 2025 systematically evaluated seven target prediction methods using a shared benchmark dataset of FDA-approved drugs [6]. The table below summarizes the quantitative performance data and key characteristics of these representative methods, providing an objective basis for comparison.
Table 1: Performance and characteristics of representative DTI prediction methods
| Method | Type | Algorithm | Key Data Source | Performance Notes |
|---|---|---|---|---|
| MolTarPred [6] | Ligand-centric | 2D similarity | ChEMBL 20 | Most effective method in 2025 comparison; Morgan fingerprints with Tanimoto score performed best. |
| PPB2 [6] | Ligand-centric | Nearest Neighbor/Naïve Bayes/Deep Neural Network | ChEMBL 22 | Uses top 2000 similar ligands; integrates multiple algorithms. |
| RF-QSAR [6] | Target-centric | Random Forest | ChEMBL 20 & 21 | ECFP4 fingerprints; considers top 4 to 110 similar ligands. |
| TargetNet [6] | Target-centric | Naïve Bayes | BindingDB | Utilizes multiple fingerprints (FP2, MACCS, ECFP). |
| ChEMBL [6] | Target-centric | Random Forest | ChEMBL 24 | Uses Morgan fingerprints. |
| CMTNN [6] | Target-centric | Multitask Neural Network | ChEMBL 34 | Run locally via ONNX runtime. |
| SuperPred [6] | Ligand-centric | 2D/Fragment/3D Similarity | ChEMBL & BindingDB | Employs ECFP4 fingerprints. |
Beyond this direct comparison, other machine learning approaches demonstrate the potential of handling highly imbalanced datasets, a common challenge in DTI prediction where known interactions are vastly outnumbered by unknown pairs. A 2025 study using a random forest classifier combined with an undersampling strategy (NearMiss) achieved state-of-the-art performance on gold standard datasets, with auROC values of up to 99.33% on enzymes and 92.26% on nuclear receptors [14]. Furthermore, novel deep learning models like DrugMAN, which integrate heterogeneous network information using mutual attention networks, have shown robust performance, particularly in challenging "cold-start" scenarios where information about new drugs or targets is limited [15].
Experimental Protocols for Method Evaluation
To ensure the reliability and reproducibility of DTI prediction methods, rigorous experimental protocols are essential. The following sections detail the key methodologies for benchmark dataset preparation and model evaluation as employed in recent comparative studies.
Benchmark Dataset Preparation
A critical first step involves constructing a high-quality, non-overlapping benchmark dataset. The following protocol is adapted from the 2025 comparative study [6]:
- Database Selection and Hosting: Select a comprehensive, experimentally validated database such as ChEMBL (version 34). Host the PostgreSQL version of the database locally for efficient data retrieval.
- Data Retrieval and Filtering: Query relevant tables (
molecule_dictionary,target_dictionary,activities) to retrieve bioactivity records. Filter for records with standard values (IC50, Ki, or EC50) below a specific threshold (e.g., 10,000 nM) to ensure high activity. - Data Cleaning:
- Exclude entries associated with non-specific or multi-protein targets by filtering out target names containing keywords like "multiple" or "complex."
- Remove duplicate compound-target pairs, retaining only unique interactions.
- High-Confidence Filtering (Optional): To enhance data quality for specific analyses, create a filtered subset that only includes interactions with a high confidence score (e.g., a minimum score of 7 in ChEMBL, indicating direct protein complex subunits are assigned).
- Benchmark Set Creation for Evaluation:
- Collect molecules with FDA approval years from the database.
- To prevent bias and overestimation of performance, ensure these molecules are excluded from the main database used for prediction.
- Randomly select a sample (e.g., 100 FDA-approved drugs) to serve as the query set for validation.
This workflow is visualized below, outlining the sequence of steps from raw data to a finalized benchmark dataset.
Performance Evaluation Strategy
The evaluation of different DTI prediction methods should be designed to simulate real-world scenarios and test generalization ability. The protocol used by the DrugMAN study exemplifies this approach [15]:
- Scenario-Based Data Splitting: Partition the known drug-target pairs into four different settings to assess model robustness:
- Warm Start: Both drugs and targets in the test set have known interactions in the training data.
- Drug-Cold: Drugs in the test set are new (absent from the training data).
- Target-Cold: Targets in the test set are new.
- Both-Cold: Both drugs and targets in the test set are new.
- Model Training and Prediction: Train each model on the training set and generate interaction predictions for the test set.
- Performance Metrics Calculation: Calculate standard metrics including the Area Under the Receiver Operating Characteristic Curve (AUROC), the Area Under the Precision-Recall Curve (AUPRC), and the F1-score. A model with good generalization will show the smallest decrease in these metrics from warm-start to cold-start scenarios.
Successful DTI prediction relies on a suite of computational tools and databases. The table below catalogs key resources, their primary functions, and their relevance to the discussed methodologies.
Table 2: Key research reagents and resources for DTI prediction
| Resource Name | Type | Function | Relevance to Methodologies |
|---|---|---|---|
| ChEMBL [6] [16] | Database | Provides experimentally validated bioactivity data (IC50, Ki, etc.) for compounds and targets. | Primary data source for ligand-based and PCM methods. |
| BindingDB [6] [16] | Database | Focuses on measured binding affinities between drugs and targets. | Key resource for ligand-based and target-centric models. |
| PDBbind [16] | Database | A curated subset of the PDB with binding affinity data for protein-ligand complexes. | Essential for training and testing structure-based, target-centric methods. |
| DrugBank [12] [15] | Database | Contains comprehensive drug and drug-target information. | Used for benchmarking and validating predictions, especially for approved drugs. |
| AlphaFold [13] | Tool | Predicts high-accuracy 3D protein structures from amino acid sequences. | Expands target coverage for structure-based docking by providing reliable models for proteins without experimental structures. |
| Molecular Fingerprints (e.g., Morgan, ECFP) [6] | Molecular Descriptor | Encodes molecular structure into a fixed-length bit string for similarity search and machine learning. | Core component of ligand-based methods and feature input for many machine learning models. |
| BindingNet v2 [16] | Dataset | A large collection of 689,796 modeled protein-ligand complexes. | Augments training data for deep learning models, improving generalization for novel ligand prediction. |
| NearMiss [14] | Algorithm | An undersampling strategy to balance imbalanced datasets by controlling the number of majority class samples. | Used in data preprocessing to improve model performance on datasets with few known interactions. |
| Mutual Attention Network [15] | Algorithm | A deep learning component that captures interaction information between drug and target representations. | Enhances hybrid and network-based models by learning complex, non-linear interaction patterns. |
The comparative analysis presented in this guide demonstrates that both ligand-based and target-based chemogenomic approaches offer distinct advantages. Ligand-centric methods like MolTarPred excel in efficiency and are highly effective for target fishing and drug repurposing when similar ligands are known [6]. In contrast, target-centric methods provide a mechanistic basis for interaction based on structural complementarity, which is crucial for novel target screening [13] [10]. The emerging trend, however, points toward hybridization and data integration. Methods that combine ligand and target information, such as proteochemometric modeling [12], or that leverage heterogeneous network data with advanced deep learning architectures, such as DrugMAN [15], show superior generalization ability, especially in challenging cold-start scenarios. Furthermore, the availability of larger and more diverse datasets, like BindingNet v2, and the integration of high-quality predicted structures from AlphaFold, are set to further enhance the accuracy and scope of all computational DTI prediction methods, solidifying their role as indispensable tools in modern drug discovery [13] [16].
The Shift from Single-Target to Polypharmacology and Systems-Level Analysis
For much of the past century, drug discovery was dominated by a "one target–one drug" approach, focused on developing highly selective ligands for individual disease proteins based on the belief that this would maximize therapeutic benefit and minimize off-target effects [17]. While this strategy achieved some successes, it has major limitations: approximately 90% of single-target drug candidates fail in late-stage trials due to lack of efficacy or unexpected toxicity [17]. These failures often stem from the reductionist oversight of the complex, redundant, and networked nature of human biology, where targeting a lone node in a complex network can easily be circumvented by the system [17].
In contrast, polypharmacology represents a fundamental paradigm shift—the rational design of small molecules that act on multiple therapeutic targets simultaneously [17]. This approach offers a transformative strategy to overcome biological redundancy, network compensation, and drug resistance, particularly for complex diseases with multifactorial etiologies [17] [18]. The clinical success of many "promiscuous" drugs revealed that multi-target activity could be advantageous rather than detrimental, leading to the intentional development of multi-target-directed ligands (MTDLs) [17] [18]. Polypharmacology has demonstrated particular success across oncology, neurodegeneration, metabolic disorders, and infectious diseases, where synergistic therapeutic effects can be achieved while potentially reducing adverse events and improving patient compliance compared to combination therapies [17].
Underpinning this shift are advanced computational methods that enable systematic analysis at a systems level. Chemogenomics—the systematic screening of targeted chemical libraries against families of drug targets—has emerged as a key strategy that integrates target and drug discovery [19] [2]. This approach leverages both ligand-based and target-based methods to efficiently explore chemical and target space, accelerating the identification of novel therapeutic targets and bioactive compounds [2] [11].
Computational Methodologies: A Comparative Framework
Computational approaches for target identification and drug discovery have evolved into two primary categories: ligand-based and target-based methods, both of which can be applied within a chemogenomics framework.
Ligand-Based Approaches
Ligand-based methods rely on the similarity principle: molecules with similar structural features are likely to exhibit similar biological activities [20]. These approaches are particularly valuable when the 3D structure of the target protein is unknown.
Similarity-Based Virtual Screening: This technique identifies new hits from large compound libraries by comparing candidate molecules against known active compounds using 2D molecular fingerprints or 3D molecular descriptors [20]. The underlying assumption is that structurally similar molecules will interact with similar biological targets.
Quantitative Structure-Activity Relationship (QSAR) Modeling: QSAR uses statistical and machine learning methods to relate molecular descriptors to biological activity [20]. Both 2D and 3D QSAR models are used for virtual screening and compound prioritization, with recent advances in 3D QSAR methods improving predictive accuracy even with limited data.
Target-Based Approaches
Target-based methods utilize the 3D structural information of target proteins to predict interactions with potential drug candidates.
Molecular Docking: This core technique predicts the bound orientation and conformation of ligand molecules within a target's binding pocket, scoring them based on interaction energies [20]. Docking performs flexible ligand docking while often treating proteins as rigid, a simplification that enables high-throughput screening.
Free-Energy Perturbation (FEP): A more computationally intensive method that estimates binding free energies using thermodynamic cycles [20]. FEP is primarily used during lead optimization to quantitatively evaluate the impact of small structural modifications on binding affinity.
Hybrid and Integrated Approaches
Increasingly, researchers are combining ligand-based and target-based methods to leverage their complementary strengths:
Sequential Integration: Large compound libraries are first filtered using fast ligand-based screening, after which the most promising candidates undergo more computationally intensive structure-based methods like docking [20].
Parallel Screening: Both approaches are run independently on the same compound library, with results compared or combined in a consensus scoring framework to increase confidence in selecting true positives [20].
Complementary Information Capture: Ensembles of protein pocket conformations can capture binding site flexibility, while ligand-based methods infer critical binding features from known active molecules [20].
The following diagram illustrates the workflow for integrating these complementary approaches:
Performance Comparison of Target Prediction Methods
Systematic Evaluation of Prediction Tools
A precise comparison study evaluated seven target prediction methods using a shared benchmark dataset of FDA-approved drugs to ensure reliability and consistency across different approaches [6]. The study assessed both stand-alone codes and web servers, including MolTarPred, PPB2, RF-QSAR, TargetNet, ChEMBL, CMTNN, and SuperPred [6]. The evaluation used ChEMBL version 34, containing 15,598 targets, 2.4 million compounds, and 20.7 million interactions, with data quality enhanced by filtering for high-confidence interactions [6].
Table 1: Comparative Performance of Target Prediction Methods
| Method | Type | Algorithm | Fingerprints/Descriptors | Key Findings |
|---|---|---|---|---|
| MolTarPred | Ligand-centric | 2D similarity | MACCS, Morgan | Most effective method overall; Morgan fingerprints with Tanimoto scores outperformed MACCS [6] |
| PPB2 | Ligand-centric | Nearest neighbor/Naïve Bayes/deep neural network | MQN, Xfp, ECFP4 | Uses top 2000 similar ligands for prediction [6] |
| RF-QSAR | Target-centric | Random forest | ECFP4 | Considers top 4, 7, 11, 33, 66, 88 and 110 similar ligands [6] |
| TargetNet | Target-centric | Naïve Bayes | FP2, Daylight-like, MACCS, E-state, ECFP2/4/6 | Algorithm uses multiple fingerprint types [6] |
| ChEMBL | Target-centric | Random forest | Morgan | Web server implementation [6] |
| CMTNN | Target-centric | ONNX runtime | Morgan | Stand-alone code implementation [6] |
| SuperPred | Ligand-centric | 2D/fragment/3D similarity | ECFP4 | Integrates similarity approaches [6] |
Performance Optimization Strategies
The comparative analysis revealed several key optimization strategies for improving prediction accuracy:
Fingerprint and Metric Selection: For MolTarPred, Morgan fingerprints with Tanimoto similarity scores demonstrated superior performance compared to MACCS fingerprints with Dice scores [6]. This highlights the importance of fingerprint and metric selection in optimizing ligand-based prediction methods.
High-Confidence Filtering: Employing high-confidence filtering (minimum confidence score of 7 in ChEMBL database) improved data quality but reduced recall, making this approach less ideal for drug repurposing applications where maximizing potential hit identification is prioritized [6].
Database Selection: ChEMBL was identified as particularly suitable for novel protein target prediction due to its extensive chemogenomic data, while DrugBank proved more appropriate for predicting new drug indications against known targets [6].
Experimental Protocols and Methodologies
Benchmark Dataset Preparation
The experimental protocol for comparative evaluation of target prediction methods involves carefully constructed benchmark datasets and validation frameworks:
Data Source and Curation: Researchers utilized ChEMBL version 34, containing over 2.4 million compounds and 20.7 million interactions [6]. The database was queried locally via PostgreSQL, retrieving data from moleculedictionary, targetdictionary, and activities tables.
Bioactivity Criteria: Records were selected with standard values for IC50, Ki, or EC50 below 10000 nM to ensure relevant binding affinities [6]. To prevent redundancy, duplicate compound-target pairs were removed, retaining only unique pairs and resulting in 1,150,487 unique ligand-target interactions.
Quality Control Measures: Entries associated with non-specific or multi-protein targets were excluded by filtering out targets containing keywords like "multiple" or "complex" [6]. A filtered database containing only high-confidence interactions (minimum confidence score of 7) was employed to enhance data quality.
Benchmark Construction: Molecules with FDA approval years were collected to prepare a benchmark dataset of FDA-approved drugs [6]. To prevent bias, these molecules were excluded from the main database to avoid overlap with known drugs during prediction. A random selection of 100 samples from the FDA-approved drugs dataset was used for method validation.
Method Implementation and Evaluation
Tool Execution: The seven target prediction methods included both stand-alone codes (MolTarPred and CMTNN) run locally and web servers (PPB2, RF-QSAR, TargetNet, ChEMBL, and SuperPred) that required manual query [6].
Performance Metrics: Methods were evaluated based on recall and prediction accuracy, with specific attention to how optimization strategies like high-confidence filtering affected performance characteristics [6].
Research Reagent Solutions for Chemogenomic Studies
Table 2: Essential Research Reagents and Databases for Chemogenomics
| Resource | Type | Primary Application | Key Features |
|---|---|---|---|
| ChEMBL | Database | Bioactivity data repository | 2.4M+ compounds, 15.5K+ targets, 20.7M+ interactions; experimentally validated data [6] |
| MolTarPred | Software Tool | Target prediction | Ligand-centric; uses Morgan fingerprints with Tanimoto similarity; top performer in comparative study [6] |
| PPB2 | Web Server | Polypharmacology prediction | Combines nearest neighbor, Naïve Bayes, and deep neural networks; uses MQN, Xfp, ECFP4 fingerprints [6] |
| RF-QSAR | Web Server | Target prediction | Random forest algorithm with ECFP4 fingerprints; target-centric approach [6] |
| AutoDock | Software Tool | Molecular docking | Models flexibility in targeted macromolecules; improved free-energy scoring system [11] |
| AlphaFold | AI Tool | Protein structure prediction | Generates high-quality structural models from amino acid sequences [17] |
Case Studies and Practical Applications
Successful Drug Repurposing through Target Prediction
Fenofibric Acid: A case study on fenofibric acid demonstrated its potential for drug repurposing as a THRB modulator for thyroid cancer treatment using computational target prediction approaches [6]. This illustrates the practical application of these methods in identifying new therapeutic uses for existing drugs.
Gleevec (imatinib mesylate): Originally developed for leukemia targeting Bcr-Abl fusion gene, Gleevec was later found to interact with PDGF and KIT receptors, leading to its repurposing for gastrointestinal stromal tumors [11]. This successful repositioning story highlights how single drugs can interact with multiple targets, enriching their polypharmacology.
Mebendazole and Actarit: MolTarPred discovered hMAPK14 as a potent target of mebendazole, confirmed by in vitro validation [6]. Similarly, the method predicted Carbonic Anhydrase II (CAII) as a new target of Actarit, suggesting potential repurposing of this rheumatoid arthritis drug for hypertension, epilepsy, and certain cancers [6].
Emerging Trends and Future Directions
The field of polypharmacology and chemogenomics continues to evolve with several emerging trends:
AI-Driven Polypharmacology: Recent advances in deep learning, reinforcement learning, and generative models have accelerated the discovery and optimization of multi-target agents [17]. These AI-driven platforms enable de novo design of dual and multi-target compounds, some demonstrating biological efficacy in vitro.
Integration of Omics Data: Combining chemogenomic approaches with CRISPR functional screens, pathway simulations, and systems biology provides a more comprehensive framework for guiding multi-target design [17].
Clinical Translation: Recent drug approvals continue to reflect the polypharmacology trend. Among 73 new drugs introduced in 2023-2024 in Germany, 18 (approximately 25%) aligned with polypharmacology concepts, including 10 antitumor agents, 5 drugs for autoimmune disorders, and 1 antidiabetic/anti-obesity drug [18].
The following diagram illustrates how polypharmacology addresses the limitations of single-target approaches in complex disease networks:
The shift from single-target to polypharmacology represents a fundamental evolution in drug discovery, moving from reductionist approaches to systems-level analysis that acknowledges the complex, networked nature of biological systems and disease pathologies. Computational methods have been instrumental in enabling this transition, with ligand-based and target-based chemogenomic approaches providing complementary tools for identifying and optimizing multi-target-directed ligands.
Performance comparisons reveal that integrated approaches leveraging both ligand and target information typically outperform single-method strategies, with tools like MolTarPred demonstrating particular efficacy when properly optimized. As artificial intelligence and machine learning continue to advance, alongside growing chemical and biological databases, the systematic discovery and optimization of polypharmacological agents is poised to become increasingly sophisticated and effective.
For researchers and drug development professionals, the practical implication is clear: embracing polypharmacology through integrated computational methods can address the limitations of single-target strategies, potentially leading to more effective therapeutics for complex diseases that have proven resistant to traditional approaches. The continued development and refinement of these computational tools will be essential for realizing the full potential of polypharmacology in delivering next-generation medicines.
Chemogenomics represents a systematic approach to understanding the interactions between chemical compounds and biological targets on a genomic scale. This field relies heavily on robust, well-curated public databases to accelerate drug discovery by enabling the prediction and analysis of Drug-Target Interactions (DTIs). The high costs, extended timelines (typically 10-15 years), and low success rates (approximately 6.3% as of 2022) of traditional drug development have made in silico approaches using these databases indispensable for preliminary screening and target identification [13]. These computational methods efficiently leverage the growing amount of available bioactivity and structural data to mitigate the risks and resource demands of experimental validation alone. Within this landscape, three databases have emerged as fundamental resources: ChEMBL for bioactivity data, the Protein Data Bank (PDB) for structural biology, and DrugBank for pharmaceutical knowledge. This guide provides an objective comparison of these resources, framing their capabilities within the context of ligand-based versus target-based chemogenomic approaches, and delivers practical experimental protocols for their application in predictive research.
Table 1: Core Characteristics of Major Chemogenomic Databases
| Feature | ChEMBL | DrugBank | RCSB PDB |
|---|---|---|---|
| Primary Focus | Bioactive molecules with drug-like properties & quantitative bioactivity [21] | Comprehensive drug & drug target information, including mechanistic & pharmacological data [22] [23] | Experimentally-determined 3D structures of biological macromolecules [24] |
| Key Data Types | Bioactivity measurements (e.g., IC50, Ki), targets, assays, molecules, documents [25] | Drug structures, mechanisms of action, interactions (drug-drug, drug-food), pharmacokinetics, targets [26] [22] | Atomic coordinates, experimental data, computed structure models, ligands, nucleic acids, and complexes [24] [27] |
| Data Source | Manually curated from literature, direct depositions, other public databases [25] | Manually curated from textbooks, journal articles, and external databases [23] | Experimental methods (X-ray, NMR, Cryo-EM) deposited by researchers; CSMs from AlphaFold DB [24] [27] |
| Unique Identifier | ChEMBL ID (e.g., 'CHEMBL123456') [25] | DrugBank ID (e.g., 'DB00001') | PDB ID (e.g., '1ABC') [24] |
| Quantitative Strength | pChEMBL value for standardized potency/affinity comparison [25] | FDA approval status, drug interactions, dosing information [22] | Resolution, R-factor, clustering data for structure quality [24] |
Database-Specific Capabilities and Research Applications
ChEMBL: A Repository for Bioactivity Data and Ligand-Based Discovery
ChEMBL is a manually curated database of bioactive molecules with drug-like properties, originally launched in 2009 [21] [25]. Its core function is to store quantitative bioactivity data (e.g., IC50, Ki) extracted from scientific literature, which is then standardized into searchable and comparable formats like the pChEMBL value—a negative logarithmic scale that allows for the comparison of roughly comparable measures of half-maximal response concentration, potency, or affinity [25]. The database has significantly expanded from its initial focus on medicinal chemistry literature to include data from patents, direct depositions from academic and industrial groups, and integrated datasets from other public resources like PubChem BioAssay [25]. This makes ChEMBL an essential tool for ligand-based virtual screening methods, such as Quantitative Structure-Activity Relationship (QSAR) modeling and pharmacophore modeling, which predict new drug candidates by leveraging known bioactivity data from structurally similar compounds [13].
DrugBank: Integrating Drug and Target Knowledge for Pharmaceutical Research
DrugBank is a unique bioinformatics and cheminformatics resource that combines detailed chemical, pharmacological, and pharmaceutical drug data with comprehensive drug target information [22] [23]. First released in 2006, it serves as a "gold standard" knowledgebase that bridges the gap between discrete chemical data and biological target contexts [22] [23]. Its DrugCard entries provide extensive information, including drug indications, mechanisms of action, biotransformation pathways, drug-drug interactions, and sequences and structures of protein targets [23]. This integrated view is particularly valuable for understanding polypharmacology, identifying drug repurposing opportunities, and predicting off-target effects. The database is crucial for research that requires a holistic understanding of a drug's pharmacological profile, from its chemical structure to its clinical applications and metabolizing enzymes [22].
Protein Data Bank (PDB): The Structural Foundation for Target-Based Approaches
The RCSB Protein Data Bank is the global archive for experimentally-determined 3D structures of biological macromolecules, including proteins, nucleic acids, and complex assemblies [24] [27]. Established in 1971, the PDB provides the foundational structural data required for target-based drug discovery approaches [27]. Its primary value lies in enabling structure-based drug design, most notably through molecular docking, a technique that uses the 3D structure of target proteins to position candidate drug molecules within active sites and simulate potential binding interactions [13]. A key recent development is the integration of over one million Computed Structure Models (CSMs) from AlphaFold DB and ModelArchive, which provide predictive models for proteins without experimentally-solved structures, thereby greatly expanding the structural coverage of the proteome [24] [27]. The PDB is also committed to open access and education, with resources like the "Molecule of the Month" series making structural biology accessible to a broad audience [27].
Experimental Protocols for Chemogenomic Workflows
The following protocols outline standard methodologies for leveraging these databases in ligand-based and target-based prediction tasks, which represent the two major computational paradigms in chemogenomics.
Protocol 1: Ligand-Based Virtual Screening Using ChEMBL
This protocol uses known active compounds from ChEMBL to identify new candidates without requiring a 3D protein structure.
Methodology:
- Define Query and Extract Actives: Identify a target of interest (e.g., a specific kinase). Query the ChEMBL database to extract all compounds reported with bioactivity (e.g., IC50 < 1 µM) against that target, along with their associated canonical SMILES strings and pChEMBL values [25].
- Calculate Molecular Descriptors: For each active compound, compute a set of molecular descriptors (e.g., molecular weight, LogP, topological polar surface area) and molecular fingerprints (e.g., ECFP4 fingerprints) using a cheminformatics toolkit like RDKit.
- Build a Predictive Model:
- QSAR Model: Use the computed descriptors and associated bioactivity values (pChEMBL) to train a regression model (e.g., Random Forest or Support Vector Machine) to predict the activity of new compounds [13].
- Similarity Search: Use the molecular fingerprints of the known actives to screen a large virtual compound library (e.g., the ZINC database). Compounds with a Tanimoto coefficient above a defined threshold (e.g., 0.7) are considered potential hits.
- Validate and Prioritize Hits: Use internal cross-validation to assess model performance. Apply the model or similarity search to an external test set from a later ChEMBL release to validate its predictive power. Prioritize compounds with high predicted activity or similarity for further experimental testing.
Protocol 2: Target-Based Screening Using Molecular Docking with PDB Structures
This protocol predicts binding modes and affinities by leveraging the 3D structural information from the PDB.
Methodology:
- Target Selection and Preparation: Search the RCSB PDB for a high-resolution structure of your target protein, ideally in complex with a relevant small molecule [24]. Prioritize structures with high resolution (e.g., < 2.0 Å) and completeness. Prepare the protein structure by removing water molecules (except those involved in key interactions), adding hydrogen atoms, and assigning correct protonation states.
- Ligand Library Preparation: Obtain a library of candidate compounds, for example, investigational drugs from DrugBank [26]. Generate 3D conformers for each compound and optimize their geometries using energy minimization.
- Molecular Docking: Define the binding site on the protein target, typically based on the location of a co-crystallized ligand. Dock each compound from your library into the binding site using software like AutoDock Vina or Glide. The docking algorithm will score and rank poses based on estimated binding free energy [13].
- Analysis and Hit Selection: Analyze the top-ranking poses for key interactions with the protein target, such as hydrogen bonds, hydrophobic contacts, and salt bridges. Visually inspect the most promising complexes. Select compounds with favorable docking scores and plausible interaction geometries for experimental validation.
The logical flow of these two primary approaches within a research project can be visualized as parallel pathways that inform each other.
Comparative Analysis: Ligand-Based vs. Target-Based Data Utilization
The choice between ligand-based and target-based approaches often depends on the available data, which in turn dictates the primary database used. The following table contrasts how each database supports these distinct yet complementary strategies.
Table 2: Database Support for Ligand-Based vs. Target-Based Approaches
| Aspect | Ligand-Based (ChEMBL-Centric) | Target-Based (PDB-Centric) |
|---|---|---|
| Primary Database | ChEMBL | RCSB PDB |
| Core Data Used | Bioactivity values (IC50, Ki), chemical structures, SMILES strings [13] [25] | 3D atomic coordinates of protein-ligand complexes [13] |
| Key Predictive Method | QSAR, Pharmacophore Modeling, Similarity Search [13] | Molecular Docking, Structure-Based Virtual Screening [13] |
| Primary Advantage | Does not require a 3D protein structure; can leverage vast historical bioactivity data [13] | Provides atomic-level insight into binding modes and interactions; can design novel scaffolds [13] |
| Key Limitation | Limited to chemical space similar to known actives; struggles with novel targets (cold-start) [13] | Highly dependent on the availability and quality of a 3D structure; scoring functions can be inaccurate [13] |
| Role of DrugBank | Provides clinical context, mechanisms of action, and known drug compounds for library building [23] | Offers links between approved/experimental drugs and their known protein targets for validation [22] |
Emerging Trends and Integrated Approaches
The distinction between ligand-based and target-based methods is increasingly blurred by modern integrated and machine learning approaches. For instance, the DGraphDTA model constructs protein graphs based on protein contact maps derived from 3D structures in the PDB, thereby incorporating spatial information into affinity prediction [13]. Furthermore, the application of Large Language Models (LLMs) and the integration of AlphaFold predicted models are advancing feature engineering for targets without experimental structures [13]. These hybrid methods, which often pull data from all three databases, aim to overcome the inherent limitations of any single approach, such as data sparsity and the "cold-start" problem for new targets with no known binders [13].
The Scientist's Toolkit: Essential Research Reagent Solutions
Successful in silico chemogenomic research relies on a digital toolkit of software and resources that facilitate the extraction, processing, and analysis of data from these primary databases.
Table 3: Essential Digital Tools for Chemogenomic Research
| Tool/Resource | Function | Application Context |
|---|---|---|
| RDKit | An open-source cheminformatics toolkit for descriptor calculation, fingerprint generation, and molecular operations. | Critical for preparing ligand libraries from ChEMBL or DrugBank for QSAR and docking studies [13]. |
| AutoDock Vina | A widely used molecular docking program for predicting protein-ligand binding poses and scoring affinities. | The standard software for target-based virtual screening using structures from the PDB [13]. |
| RCSB PDB API | A programmatic interface to search, filter, and retrieve data and structures from the PDB archive. | Enables automated workflows for fetching structural data and metadata for large-scale analysis [24]. |
| pChEMBL Value | A standardized negative logarithmic scale for bioactivity data. | Allows for direct comparison of potency/affinity measurements from different assay types within ChEMBL [25]. |
| AlphaFold DB | A database of highly accurate predicted protein structures generated by DeepMind's AlphaFold2. | Provides computed structure models for targets lacking experimental structures, expanding the scope of structure-based design [24] [27]. |
In Silico Toolkits: Methods, Algorithms, and Real-World Applications
In the field of chemogenomics, drug-target interaction prediction is primarily pursued through two complementary paradigms: target-based and ligand-based approaches. Target-based methods rely on knowledge of the 3D structure of the protein target, using techniques such as molecular docking to identify potential binders [6] [28]. In contrast, ligand-based methods leverage the principle that chemically similar molecules are likely to exhibit similar biological activities [29]. This guide focuses on the latter, providing a comparative analysis of the three core ligand-based techniques: molecular similarity, Quantitative Structure-Activity Relationship (QSAR) modeling, and pharmacophore modeling. These methods are indispensable when the target protein structure is unknown, poorly characterized, or difficult to model, as is common with many membrane protein targets [28]. The fundamental advantage of ligand-based design is its ability to bypass the need for structural target information, instead using known bioactive molecules as templates to discover new hits or optimize lead compounds [6].
Comparative Performance of Ligand-Based Methods
The following table summarizes the core principles, strengths, and limitations of the three primary ligand-based methods, providing a framework for selection based on research objectives.
| Method | Core Principle | Typical Applications | Key Strengths | Primary Limitations |
|---|---|---|---|---|
| Molecular Similarity | Measures structural or property resemblance between molecules using fingerprints and similarity metrics [6] [30]. | Virtual screening, target fishing, drug repurposing [6]. | Fast, intuitive, no required activity data; effective for identifying novel chemotypes via scaffold hopping [6] [30]. | Limited by known ligand data; may miss novel chemotypes if similarity is narrowly defined [6]. |
| QSAR Modeling | Correlates numerical molecular descriptors with biological activity using statistical or ML models [31] [32]. | Lead optimization, activity and ADMET property prediction [33] [31]. | Predictive and quantitative; provides mechanistic insights; can model complex, non-linear relationships with modern ML [31]. | Requires a dataset of compounds with known activity; model performance depends on data quality and applicability domain [33]. |
| Pharmacophore Modeling | Identifies and maps the essential steric and electronic features for biological activity [34] [32]. | Virtual screening, de novo molecular generation, understanding key binding interactions [34] [32]. | Provides an intuitive 3D visualization of interaction requirements; can integrate both ligand and target structure information [34]. | Conformational analysis can be complex; quality depends on the input molecules' quality and diversity [32]. |
Performance Benchmarks and Data
Recent comparative studies provide quantitative performance data for these methods. A 2025 systematic comparison of target prediction methods evaluated several ligand-centric approaches using a benchmark of FDA-approved drugs [6]. The study found that MolTarPred, a 2D similarity-based method, was the most effective for drug repurposing, with optimizations showing that Morgan fingerprints with a Tanimoto similarity score outperformed other fingerprint and metric combinations [6].
For QSAR models, best practices are evolving. A 2025 analysis demonstrated that for virtual screening of ultra-large chemical libraries, models trained on imbalanced datasets (reflecting the real-world abundance of inactives) and optimized for high Positive Predictive Value (PPV) achieved hit rates at least 30% higher than models trained on balanced datasets and optimized for Balanced Accuracy [35]. This highlights the critical importance of selecting the right performance metric for the specific application.
Experimental Protocols and Methodologies
Molecular Similarity Search Protocol
Objective: To identify potential inhibitors of a target protein using a known active compound as a query. Workflow:
- Query Selection: Select a known high-affinity ligand (e.g., from ChEMBL or BindingDB) and obtain its canonical SMILES string [6].
- Fingerprint Generation: Encode the query molecule and all molecules in the screening database (e.g., ZINC, ChEMBL) into a molecular fingerprint. The Morgan fingerprint (radius 2, 2048 bits) is a current recommended choice [6].
- Similarity Calculation: Compute the similarity between the query fingerprint and every database fingerprint using the Tanimoto coefficient [6].
- Hit Identification: Rank all database compounds by their similarity score and select the top candidates for further experimental validation [6].
QSAR Model Development Protocol
Objective: To build a predictive model that relates molecular structures to a specific biological activity (e.g., pIC50). Workflow:
- Data Curation: Collect a set of compounds with reliably measured biological activity. A minimum of 20-30 compounds is recommended [32].
- Descriptor Calculation: Use software like PaDEL or RDKit to compute numerical molecular descriptors (e.g., molecular weight, logP, topological indices) for all compounds [31] [32].
- Dataset Splitting: Divide the data into a training set (~80%) for model building and a test set (~20%) for validation [32].
- Model Training: Use the training set and machine learning algorithms (e.g., Random Forest, Support Vector Machines) to build a model that correlates descriptors with activity [31].
- Model Validation: Critically assess the model's predictive power on the held-out test set using metrics like R² (for continuous models) and PPV (for classification models used in virtual screening) [35].
Ligand-Based Pharmacophore Modeling Protocol
Objective: To create a 3D model of the essential features a molecule must possess to bind to a target, using a set of known active ligands. Workflow (as applied to dengue protease inhibitors [32]):
- Ligand Selection: Select a set of 3-5 known active compounds with diverse structures but high potency. Energy-minimize their structures using a tool like Avogadro with the MMFF94 force field [32].
- Model Generation: Input the energy-minimized structures (in
.mol2format) into pharmacophore modeling software like PharmaGist. The software will identify common chemical features (e.g., hydrogen bond donors/acceptors, hydrophobic regions, aromatic rings) and their spatial arrangement [32]. - Model Screening: Use the resulting pharmacophore hypothesis to screen a large database (e.g., ZINC) via a tool like ZINCPharmer to find molecules that match the feature arrangement [32].
- Hit Refinement: Subject the virtual hits to further analysis, such as molecular docking and ADMET prediction, to prioritize the most promising candidates for experimental testing [32].
Figure 1: A workflow diagram illustrating the three primary ligand-based methodologies for computational drug discovery. All paths begin with known active ligands and converge on the identification of new candidate compounds for experimental testing.
The following table details key computational tools, databases, and descriptors essential for implementing the ligand-based methods discussed.
| Tool/Resource Name | Type | Primary Function | Relevance to Methods |
|---|---|---|---|
| ChEMBL [6] | Database | Curated database of bioactive molecules with drug-like properties. | Source of known active ligands for all three methods. |
| ZINC [32] | Database | Publicly available database of commercially-available compounds for virtual screening. | Primary screening library for similarity and pharmacophore searches. |
| RDKit [34] | Cheminformatics Toolkit | Open-source toolkit for cheminformatics and machine learning. | Fingerprint and descriptor calculation; general molecular manipulation. |
| PaDEL [32] | Software | Calculates molecular descriptors and fingerprints. | Descriptor calculation for QSAR modeling. |
| Morgan Fingerprints [6] | Molecular Representation | A type of circular fingerprint encoding the atomic environment. | State-of-the-art fingerprint for molecular similarity calculations. |
| PharmaGist [32] | Software | Online server for generating ligand-based pharmacophore models. | Creates pharmacophore hypotheses from a set of active ligands. |
| ZINCPharmer [32] | Web Server | Online tool for screening the ZINC database using a pharmacophore query. | Identifies molecules that match a given pharmacophore model. |
| Random Forest [31] | Algorithm | Robust machine learning algorithm for classification and regression. | A top choice for building modern, non-linear QSAR models. |
Molecular similarity, QSAR, and pharmacophore modeling represent a powerful arsenal of ligand-based methods that remain crucial in modern drug discovery. The choice between them depends on the available data and the specific research goal: molecular similarity for rapid screening and repurposing, QSAR for quantitative activity prediction and lead optimization, and pharmacophore modeling for a more structural understanding of interaction requirements. The ongoing integration of artificial intelligence, through graph neural networks and transformers, is enhancing the predictive power and scope of these classical approaches [31] [30]. By understanding the comparative strengths, performance benchmarks, and experimental protocols outlined in this guide, researchers can make informed decisions to efficiently navigate the vast chemical space and accelerate the discovery of novel therapeutic agents.
Target-based computational methods are pillars of modern structure-based drug design, enabling the prediction of how small molecules interact with biological macromolecules at an atomic level. These methods, which include molecular docking, Structure-Based Virtual Screening (SBVS), and Free-Energy Perturbation (FEP), leverage 3D structural information to prioritize compounds for synthesis and experimental testing, significantly accelerating early-stage drug discovery campaigns [36] [37]. Their application is crucial for hit identification, lead optimization, and understanding polypharmacology.
This guide provides a comparative analysis of these three key methodologies, framing them within the broader context of chemogenomic approaches. While ligand-based methods rely on the principle that similar molecules have similar activities, target-based methods utilize the physical structure of the target protein, offering a powerful, mechanism-driven strategy for discovering novel scaffolds, even in the absence of known active compounds.
The three methods differ fundamentally in their computational intensity, primary application, and the qualitative versus quantitative nature of their predictions. Table 1 summarizes their core characteristics and typical performance metrics.
Table 1: Comparative Overview of Target-Based Methods
| Method | Primary Application | Computational Cost | Key Performance Metrics | Typical Performance |
|---|---|---|---|---|
| Molecular Docking | Binding pose prediction; Initial hit identification from large libraries. | Low to Moderate (GPU can accelerate) | Pose Accuracy (RMSD ≤ 2 Å); Physical Validity (PB-valid); Virtual Screening Enrichment (EF1%, logAUC) | Pose Accuracy (Traditional: >90% on known complexes; DL: >70%); EF1%: Can reach 28-31 with ML re-scoring [38] [39] |
| Structure-Based Virtual Screening (SBVS) | Prioritizing compounds from ultra-large libraries (billions of molecules). | High (scales with library size) | Hit Rate; Enrichment Factor (EF); logAUC | Hit rates improve dramatically with billion-molecule libraries; Performance is modelable and improvable [40] [41] |
| Free-Energy Perturbation (FEP) | Lead optimization; predicting binding affinity changes for congeneric series. | Very High (requires extensive GPU resources) | Mean Unsigned Error (MUE) vs. experiment; Accuracy within 1.0 kcal/mol | MUE of ~1.0 kcal/mol (6-8-fold in affinity); Successfully guides discovery of selective inhibitors [42] [37] |
Molecular docking serves as the foundational tool for predicting how a ligand binds to a protein's binding site. A comprehensive 2025 benchmarking study evaluated traditional, deep learning (DL), and hybrid docking methods across multiple dimensions [39]. The results revealed a clear performance tier for pose prediction on benchmark sets like Astex Diverse Set and PoseBusters: Traditional methods (e.g., Glide SP) and Hybrid methods (AI scoring with traditional search) lead in combined success rate (RMSD ≤ 2 Å & physically valid), followed by Generative Diffusion models (e.g., SurfDock), with Regression-based DL models trailing behind [39]. While DL methods like SurfDock can achieve high pose accuracy (>70%), they often produce physically implausible structures with steric clashes, highlighting a key limitation [39].
SBVS uses docking to search massive molecular libraries. Performance is quantified by the hit rate—the fraction of tested compounds that show activity. Studies of large-scale docking campaigns, where billions of molecules are docked and hundreds are tested, show that hit rates are a predictable function of docking score and the library's intrinsic "hit-proneness" [41]. Performance can be significantly enhanced by re-scoring docking outputs with machine learning-based scoring functions (ML SFs). For instance, re-scoring docking results for the malaria target PfDHFR with CNN-Score improved early enrichment (EF1%) from worse-than-random to 28 for the wild-type and 31 for a resistant mutant [38].
FEP provides a higher-accuracy, physics-based method for predicting relative binding free energies. It is the most computationally intensive of the three, but its predictions are highly accurate, with average errors near 1.0 kcal/mol, which is within experimental uncertainty [42] [37]. Recent advances include Absolute Binding Free Energy (ABFE) calculations, which remove the need for a closely related reference ligand, and Active Learning FEP, which combines FEP with faster QSAR methods to explore chemical space more efficiently [42]. A 2025 case study on Wee1 kinase inhibitors demonstrated FEP's power, where it was used to profile 6.7 billion design ideas, leading to the discovery of novel, potent, and selective clinical candidates [37].
Experimental Protocols and Workflows
Benchmarking Molecular Docking and SBVS Performance
A rigorous protocol for evaluating docking and SBVS performance involves using a high-quality benchmark set like DEKOIS 2.0, which contains known active molecules and structurally similar but inactive decoys [38].
Key Steps:
- Protein Preparation: Obtain crystal structures (e.g., from PDB). Remove water molecules, ions, and redundant chains. Add and optimize hydrogen atoms using tools like OpenEye's "Make Receptor" [38].
- Ligand/Decoy Preparation: Prepare the active and decoy molecules from the benchmark set. Generate multiple conformations for each ligand using software like Omega [38].
- Docking Grid Definition: Define the docking grid box centered on the binding site. The size should encompass all relevant residues (e.g., 21.33Å × 25.00Å × 19.00Å for a typical target) [38].
- Docking Execution: Perform docking with the tools being evaluated (e.g., AutoDock Vina, FRED, PLANTS) using their default search parameters [38].
- Re-scoring with ML SFs (Optional): Extract the top poses from docking and re-score them using pretrained machine learning scoring functions like CNN-Score or RF-Score-VS v2 [38].
- Performance Analysis: Calculate key metrics:
- Enrichment Factor (EF): Measures the concentration of true actives in the top-ranked fraction of the screened library. EF1% is the enrichment in the top 1% [38].
- pROC-AUC: The area under the semi-log ROC curve, assessing the method's ability to rank actives above decoys [38].
- pROC-Chemotype Plots: Analyze the diversity and affinity of actives retrieved at early enrichment stages [38].
The workflow for a comparative docking and SBVS study is visualized below.
Free-Energy Perturbation for Binding Affinity Prediction
The FEP workflow, particularly for Relative Binding Free Energy (RBFE) calculations, is based on a thermodynamic cycle that allows for the calculation of the relative binding free energy between two similar ligands without simulating the physical binding process.
Key Steps:
- System Setup: Construct the thermodynamic cycle for the ligand pair in both their protein-bound and solvated (unbound) states [42] [37].
- Lambda Schedule Selection: Define a series of intermediate, non-physical "λ states" to alchemically morph one ligand into the other. Modern tools use automated scheduling to optimize the number of windows, balancing accuracy and computational cost [42].
- Force Field Parameterization: Assign accurate parameters to describe the energetics of the system. For challenging ligand torsions, quantum mechanics (QM) calculations can be used to refine parameters [42].
- Solvation and Sampling: Ensure the system is properly hydrated. Techniques like Grand Canonical Monte Carlo (GCMC) can be used to sample water positions effectively, which is critical for reducing hysteresis [42].
- Running Simulations: Perform molecular dynamics simulations at each λ window. For perturbations involving formal charge changes, longer simulation times are typically required for reliable results [42].
- Analysis: Analyze the simulation data to compute the relative binding free energy (ΔΔG) between the two ligands. The accuracy is typically validated against experimental data, with a goal of achieving a mean unsigned error (MUE) of ~1.0 kcal/mol [37].
The following diagram illustrates the core thermodynamic cycle and key steps of an FEP workflow.
The Scientist's Toolkit: Essential Research Reagents and Databases
Successful implementation of these computational methods relies on access to robust software tools, high-quality databases, and powerful computing infrastructure. Table 2 lists key resources.
Table 2: Essential Research Reagents and Resources for Target-Based Methods
| Category | Item / Resource | Function / Description | Relevant Use Case |
|---|---|---|---|
| Software & Tools | AutoDock Vina, FRED, PLANTS | Traditional molecular docking programs. | SBVS and pose prediction benchmarking [38]. |
| Glide SP | High-performance traditional docking tool. | Known for high physical validity of poses [39]. | |
| SurfDock, DiffBindFR | Deep learning-based generative docking models. | Achieving high pose prediction accuracy [39]. | |
| CNN-Score, RF-Score-VS v2 | Machine Learning Scoring Functions. | Re-scoring docking outputs to improve virtual screening enrichment [38]. | |
| FEP Software (e.g., Flare FEP, FEP+) | Suite for running free energy calculations. | Predicting relative binding affinities during lead optimization [42] [37]. | |
| Chemprop | Deep learning framework for molecular property prediction. | Training models to predict docking scores and guide screening [40]. | |
| Databases & Benchmarks | Protein Data Bank (PDB) | Repository for experimentally determined 3D protein structures. | Source of protein structures for docking and FEP setup [38]. |
| DEKOIS 2.0 | Benchmarking sets with actives and decoys. | Evaluating docking and SBVS performance [38]. | |
| ChEMBL, BindingDB | Databases of bioactive molecules with drug-like properties, affinities, and ADMET data. | Source of known active compounds for validation and training ML models [6] [12]. | |
| Large-Scale Docking (LSD) Database | Website providing docking scores/poses for 6.3B molecules across 11 targets. | Benchmarking machine learning and chemical space exploration methods [40]. | |
| Computing Infrastructure | GPU Clusters | High-performance computing. | Essential for running FEP calculations and deep learning docking models in a practical timeframe [42] [39]. |
Molecular docking, SBVS, and FEP represent a spectrum of target-based methods with complementary strengths in drug discovery. Docking provides a fast, accessible tool for initial pose prediction and screening billions of compounds, especially when enhanced by ML re-scoring. SBVS leverages docking to efficiently explore ultra-large chemical spaces, with predictable and improvable hit rates. FEP sits at the high-fidelity end, providing quantitative, experimentally accurate affinity predictions critical for lead optimization, albeit at a higher computational cost.
The choice of method depends on the project stage and available resources. For rapid screening of vast libraries, SBVS is indispensable. For optimizing a lead series with a known binding mode, FEP offers unparalleled precision in affinity prediction. The ongoing integration of machine learning, as seen in ML scoring functions and active learning workflows, is creating powerful hybrid approaches that leverage the speed of data-driven models and the rigor of physics-based simulations. This synergy, combined with the increasing availability of high-quality protein structures from experimental methods and AI prediction tools like AlphaFold2, promises to further solidify the role of target-based methods in accelerating the discovery of new therapeutics.
The prediction of drug-target interactions (DTIs) is a cornerstone of modern drug discovery, crucial for identifying new therapeutic candidates and repurposing existing drugs [43]. Traditionally, this field has been dominated by two distinct computational philosophies: ligand-based approaches and target-based approaches. Ligand-based methods operate on the principle that similar molecules tend to interact with similar biological targets, relying heavily on the chemical fingerprint similarity of compounds [6] [11]. In contrast, target-based methods, such as molecular docking, use the three-dimensional structure of a protein target to predict how and where a small molecule might bind [11]. With the advent of robust biological databases and increased computational power, a third, more integrative paradigm has emerged: chemogenomic approaches. These methods systematically leverage both chemical and biological information, often using machine learning to learn the complex relationships between drugs and their targets from large-scale datasets [43] [11]. The evolution of machine learning (ML), from classic algorithms like Random Forests to modern deep learning and Graph Neural Networks (GNNs), has been the primary engine driving this chemogenomic revolution, leading to unprecedented accuracy in predicting novel drug-target interactions [44] [45].
Comparative Performance of Machine Learning Methods
As ML techniques have advanced, so too has their performance in DTI prediction. The following table summarizes key quantitative results from recent studies and benchmark analyses, providing a clear comparison across different algorithmic families.
Table 1: Performance Comparison of Different DTI Prediction Methods on Various Benchmark Datasets
| Method Category | Specific Model/Approach | Dataset(s) Used | Key Performance Metrics | Year/Reference |
|---|---|---|---|---|
| Similarity-Based (Ligand-Centric) | MolTarPred (Morgan fingerprints) | ChEMBL 34, FDA-approved drugs | Most effective in systematic comparison [6] | 2025 |
| Classical Machine Learning | GAN + Random Forest Classifier | BindingDB-Kd | Acc: 97.46%, AUC: 99.42% [44] | 2025 |
| Classical Machine Learning | Feature Selection + Rotation Forest | Enzyme, Ion Channels, GPCRs, Nuclear Receptors | Acc: 98.12%, 98.07%, 96.82%, 95.64% [46] | 2023 |
| Kernel & Matrix Factorization | Kernelized Bayesian Matrix Factorization | Cancer cell line screening data | Effective for integrated QSAR [46] | 2016 |
| Deep Learning (General) | DeepLPI (CNN + biLSTM) | BindingDB | AUC: 0.790 (Test Set) [44] | 2025 |
| Graph Neural Networks | Hetero-KGraphDTI (GNN + Knowledge) | Multiple benchmarks | Avg. AUC: 0.98, Avg. AUPR: 0.89 [45] | 2025 |
| Graph Neural Networks | Multi-modal GCN Framework | DrugBank | AUC: 0.96 [45] | 2023 |
| Graph Neural Networks | Graph-based Multi-network | KEGG | AUC: 0.98 [45] | 2024 |
The data reveals a clear performance trend. While classical ML models like Random Forest, especially when enhanced with feature selection [46] or data balancing techniques like Generative Adversarial Networks (GANs) [44], achieve remarkably high accuracy, modern deep learning approaches are pushing the boundaries even further. GNN-based models consistently achieve top-tier performance, with the recently proposed Hetero-KGraphDTI framework setting a new benchmark with an average AUC of 0.98 [45]. This model's integration of biological knowledge graphs appears to mitigate over-smoothing and enhance the biological interpretability of its predictions.
Experimental Protocols and Methodologies
The superior performance of modern models is a direct result of sophisticated experimental protocols and feature engineering strategies. Below is a workflow diagram illustrating a typical advanced DTI prediction pipeline, integrating elements from several state-of-the-art approaches.
Data Preparation and Feature Engineering
The foundation of any robust DTI model is high-quality, well-curated data. Commonly used databases include ChEMBL [6], BindingDB [44], and DrugBank [11], which provide experimentally validated interactions, compound structures, and target information.
- Drug Feature Extraction: Small molecules are typically represented as molecular fingerprints, such as MACCS keys or Morgan fingerprints (ECFP), which encode molecular structures as bit vectors denoting the presence or absence of specific substructures [6] [46]. Newer methods represent drugs as molecular graphs, where atoms are nodes and bonds are edges, allowing GNNs to learn informative representations directly from the graph structure [45].
- Target Feature Extraction: Proteins are represented by features derived from their amino acid sequences. Common descriptors include Amino Acid Composition (AAC), Dipeptide Composition (DC), and the Position-Specific Scoring Matrix (PSSM), which captures evolutionary information [46]. Deep learning methods can learn protein representations directly from sequence data using convolutional or recurrent neural networks [44] [45].
A critical challenge in DTI prediction is the severe class imbalance, as known positive interactions are vastly outnumbered by unknown (and typically treated as negative) pairs [44] [46]. To address this, advanced protocols employ data balancing techniques. The use of Generative Adversarial Networks (GANs) to create synthetic data for the minority class has been shown to significantly reduce false negatives and improve model sensitivity [44]. Furthermore, enhanced negative sampling strategies are crucial for graph-based models to ensure the model learns meaningful distinctions [45].
Model-Specific Workflows
Different model architectures require tailored experimental workflows, as depicted in the following diagram for a state-of-the-art Graph Neural Network approach.
- Classical ML & Feature-Based Models: As exemplified by the high-performing Rotation Forest model [46], the workflow involves extracting a wide array of features from drugs and targets, combining them into a single feature vector per drug-target pair, and then applying a feature selection algorithm (e.g., IWSSR) to reduce dimensionality and prevent overfitting. The selected features are then used to train a powerful ensemble classifier.
- Graph Neural Networks: The workflow for a model like Hetero-KGraphDTI is more complex [45]. First, a heterogeneous graph is constructed where drugs and targets are nodes, and edges represent various relationships (e.g., drug-drug similarity, target-target interaction, known DTIs). A Graph Convolutional Network (GCN) or Graph Attention Network (GAT) then performs message passing to learn node embeddings by aggregating information from neighboring nodes. A key innovation is the use of knowledge-based regularization, which incorporates information from ontologies like Gene Ontology (GO) to ensure the learned embeddings are biologically meaningful. Finally, the interaction score is computed from the final drug and target embeddings.
For researchers aiming to implement or benchmark DTI prediction models, a standardized set of computational "reagents" is essential. The following table catalogs key resources.
Table 2: Essential Computational Reagents for DTI Prediction Research
| Resource Category | Specific Resource Name | Description and Primary Function |
|---|---|---|
| Bioactivity Databases | ChEMBL [6], BindingDB [44] | Provide curated, experimentally validated drug-target interaction data for model training and benchmarking. |
| Drug Information Databases | DrugBank [11], PubChem [11] | Comprehensive repositories of drug-like molecules, their structures, and pharmacological data. |
| Target Information Databases | UniProt, Gene Ontology (GO) [45] | Provide protein sequence data, functional annotations, and pathway information for target feature extraction and knowledge integration. |
| Molecular Fingerprints | Morgan (ECFP) [6], MACCS [6] | Algorithms to convert drug molecular structures into fixed-length numerical vectors for machine learning. |
| Protein Feature Extractors | PSSM Generators, APAAC Descriptors [46] | Tools to compute evolutionarily informed and composition-based feature vectors from protein sequences. |
| Machine Learning Libraries | Scikit-learn, PyTorch, TensorFlow | Core programming libraries for implementing classical ML models, deep learning, and GNNs. |
| Graph Learning Libraries | PyTorch Geometric, Deep Graph Library (DGL) | Specialized libraries for building and training graph neural network models. |
| Validation & Benchmarking | SIDER, KEGG [45] | External datasets used for testing model generalizability and real-world performance. |
The machine learning revolution has fundamentally transformed the landscape of drug-target interaction prediction. The journey from robust, feature-based models like Random Forest to the current state-of-the-art Graph Neural Networks illustrates a clear path toward greater integration and biological fidelity. Classical models excel in contexts with well-defined, curated features and offer high interpretability [46]. In contrast, modern deep learning and GNN models leverage raw structural and sequential data to automatically learn complex representations, achieving superior performance, particularly in large-scale, data-rich environments [44] [45].
This evolution is also blurring the historical lines between ligand-based and target-based approaches. The chemogenomic philosophy is now dominant, and the most advanced models are inherently multi-modal. They do not merely use ligand similarity or target structure; they integrate chemical, structural, sequence, and network information within a unified framework [45]. The critical addition of biological knowledge through regularization techniques further ensures that predictions are not just statistically sound but also biologically plausible. As these tools continue to mature, they will increasingly serve as powerful, interpretable, and indispensable partners for researchers and scientists in accelerating the discovery of new therapeutics.
In the contemporary landscape of pharmaceutical research, drug repurposing and multi-target drug discovery have emerged as transformative paradigms that address the critical inefficiencies of traditional drug development. Whereas conventional de novo drug discovery requires an average of 10–15 years and costs exceeding $2.5 billion with failure rates of 90–95%, repurposing strategies can reduce timelines to 3–12 years at an average cost of $300 million, significantly de-risking the development process [47] [48]. This accelerated pathway leverages existing clinical compounds with established safety profiles, bypassing extensive early-stage toxicity and pharmacokinetic testing that account for many clinical failures [49] [50].
The scientific foundation underpinning these approaches is polypharmacology—the concept that small-molecule drugs often interact with multiple biological targets simultaneously, regardless of their original design intent [6] [47]. This phenomenon enables two complementary strategic approaches: drug repurposing, which identifies new therapeutic indications for existing drugs, and multi-target drug discovery, which intentionally designs compounds to modulate multiple disease-relevant pathways [6] [11]. The strategic alignment of these approaches with modern computational methods has created a powerful framework for addressing complex diseases through systematic analysis of drug-target interactions (DTIs), mechanisms of action (MoA), and network pharmacology [6] [51].
Advances in chemogenomic methodologies have been particularly instrumental in this transformation, enabling researchers to navigate the complex relationships between chemical space and biological targets through two primary computational frameworks: ligand-based approaches, which predict interactions based on chemical similarity to known active compounds, and target-based approaches, which utilize structural or sequence information about the protein targets themselves [6] [13] [51]. This article presents a comparative analysis of these methodologies through detailed case studies, performance benchmarking, and experimental protocols that highlight their respective strengths in advancing drug repurposing and multi-target drug discovery.
Methodological Framework: Ligand-Based vs. Target-Based Chemogenomic Approaches
Computational prediction of drug-target interactions forms the cornerstone of modern repurposing and polypharmacology research. The two predominant chemogenomic approaches—ligand-based and target-based—offer distinct methodological frameworks, data requirements, and analytical advantages that make them suitable for different research scenarios and resource environments.
Ligand-based methods operate on the principle that chemically similar compounds are likely to share biological targets and therapeutic effects [6] [11]. These approaches primarily utilize two-dimensional (2D) or three-dimensional (3D) chemical structure representations to calculate similarity between a query molecule and databases of known bioactive compounds with annotated targets [6]. Key techniques within this domain include similarity searching, quantitative structure-activity relationship (QSAR) modeling, and pharmacophore mapping [13] [11]. The effectiveness of ligand-based methods heavily depends on the comprehensiveness of known ligand databases and the optimal selection of molecular fingerprints (e.g., MACCS, Morgan) and similarity metrics (e.g., Tanimoto, Dice) [6]. For example, in a systematic comparison of prediction methods, MolTarPred demonstrated superior performance using Morgan fingerprints with Tanimoto scoring compared to MACCS fingerprints with Dice scoring [6].
In contrast, target-based methods leverage information about the protein targets, typically through either structure-based docking simulations or machine learning models trained on target sequence and features [6] [13] [51]. Structure-based approaches like molecular docking utilize three-dimensional protein structures to simulate how candidate drugs might bind to active sites, estimating binding affinities through computational scoring functions [13] [51]. Machine learning-based target-centric methods build predictive models for each target using various algorithms including random forest, naïve Bayes classifiers, and neural networks [6] [13]. These approaches can provide insights into binding modes and molecular interactions but traditionally depend on the availability of high-quality protein structures, a limitation increasingly addressed by computational tools like AlphaFold [6] [13].
Table 1: Comparative Analysis of Ligand-Based and Target-Based Prediction Methods
| Feature | Ligand-Based Methods | Target-Based Methods |
|---|---|---|
| Primary Data Source | Chemical structures of known ligands | Protein structures or sequences |
| Key Algorithms | 2D/3D similarity, QSAR, pharmacophore mapping | Molecular docking, random forest, neural networks |
| Representative Tools | MolTarPred, SuperPred, PPB2 | RF-QSAR, TargetNet, CMTNN, DTIAM |
| Advantages | Does not require protein structures; effective when many known ligands exist | Can predict novel targets without similar known ligands; provides mechanistic insights |
| Limitations | Limited to targets with known ligands; cannot discover truly novel target space | Dependent on protein structure availability; computationally intensive |
| Optimal Use Cases | Early-stage repurposing when chemical starting points exist | Target-centric discovery; understanding binding mechanisms |
A emerging trend in the field involves the development of hybrid frameworks that integrate both ligand and target information to overcome the limitations of individual approaches. The DTIAM framework exemplifies this trend, employing self-supervised learning on both molecular graphs of compounds and primary sequences of proteins to predict interactions, binding affinities, and mechanisms of action [51]. This unified approach has demonstrated substantial performance improvements, particularly in challenging "cold start" scenarios involving new drugs or targets with limited existing data [51].
Case Study 1: Mebendazole Repurposing in Oncology via Ligand-Based Prediction
The successful repositioning of mebendazole from an antihelminthic agent to a promising anticancer therapeutic exemplifies the power of ligand-based prediction methodologies in drug repurposing. This case study demonstrates how computational similarity searching followed by experimental validation can reveal unexpected polypharmacology with significant clinical potential.
Experimental Protocol and Workflow
The rediscovery of mebendazole began with systematic ligand-based virtual screening using the MolTarPred platform, which employs 2D chemical similarity searching against the ChEMBL database of bioactive molecules [6]. The methodological workflow followed these key stages:
Compound Selection and Fingerprint Generation: Mebendazole's chemical structure was encoded using molecular fingerprints that capture key structural features and patterns. The Morgan fingerprint with radius 2 and 2048 bits was identified as optimal for similarity calculations in comparative method evaluations [6].
Similarity Searching and Target Prediction: The algorithm calculated Tanimoto similarity scores between mebendazole and all compounds in the ChEMBL database annotated with biological targets. The top similarity hits (1, 5, 10, and 15 most similar compounds) were analyzed to identify potential shared targets [6].
Target Prioritization: The similarity-based analysis identified mitogen-activated protein kinase 14 (hMAPK14) as a high-confidence prediction based on the known targets of mebendazole's structural analogs in the database [6].
Experimental Validation: In vitro binding assays confirmed mebendazole's potent interaction with hMAPK14, validating the computational prediction and establishing a mechanistic foundation for its anticancer activity [6] [48].
The following workflow diagram illustrates this ligand-based repurposing pipeline:
Multi-Target Mechanism and Therapeutic Potential
Experimental investigation following the computational prediction revealed that mebendazole exhibits a remarkable polypharmacological profile, simultaneously targeting multiple pathways involved in oncogenesis and cancer progression [48]. Unlike targeted therapies that address single pathways, mebendazole's multi-target mechanism includes:
- Microtubule Disruption: As its primary anthelmintic mechanism, mebendazole binds to tubulin subunits in cancer cells, disrupting microtubule formation and function, which leads to cell cycle arrest and apoptosis [48].
- MAPK Pathway Inhibition: The computationally predicted interaction with hMAPK14 (p38 MAPK) was confirmed, demonstrating inhibition of this critical signaling pathway involved in cell proliferation, survival, and stress response [6] [48].
- Angiogenesis Regulation: Mebendazole suppresses tumor vascularization by inhibiting angiogenesis through modulation of HIF-1α and VEGF signaling pathways [48].
- Autophagy Modulation: The drug regulates autophagic processes in cancer cells, creating synergistic antitumor effects when combined with other therapeutics [48].
- Hedgehog Pathway Interference: Mebendazole disrupts Hedgehog signaling, which is frequently dysregulated in various cancers and associated with tumor maintenance and progression [48].
This multi-target mechanism is particularly valuable in addressing the challenge of drug resistance in oncology, as cancer cells struggle to develop simultaneous resistance across multiple pathways. The demonstrated safety profile of mebendazole from decades of clinical use for parasitic infections, combined with its efficacy across diverse tumor types, positions it as an ideal repurposing candidate with superior therapeutic index compared to conventional chemotherapeutic agents [48].
Case Study 2: DTIAM Framework for Multi-Target Prediction and MOA Analysis
The DTIAM (Drug-Target Interaction, Affinity, and Mechanism) framework represents a cutting-edge unified computational approach that advances beyond simple interaction prediction to encompass binding affinity estimation and mechanism of action (MoA) classification [51]. This case study examines its application in predicting activation/inhibition mechanisms and identifying novel TMEM16A inhibitors, demonstrating the power of integrated target-based and ligand-based methodologies.
Experimental Protocol and Workflow
DTIAM employs a sophisticated multi-module architecture that combines self-supervised pre-training with downstream prediction tasks. The experimental workflow consists of three integrated components:
Drug Molecular Pre-training Module: This module processes molecular graphs of compounds, segmenting them into substructures and learning representations through multi-task self-supervised learning. The model employs three pre-training tasks: Masked Language Modeling (recovering masked substructures), Molecular Descriptor Prediction, and Molecular Functional Group Prediction. These tasks enable the model to extract meaningful contextual information and implicit features between molecular substructures without requiring labeled interaction data [51].
Target Protein Pre-training Module: Protein sequences are processed using Transformer attention maps to learn representations and contact patterns directly from primary sequence data through unsupervised language modeling. This approach captures residue-level features and higher-order structural patterns without requiring explicit 3D structure information [51].
Unified Prediction Module: The learned representations of compounds and proteins are integrated to predict drug-target interactions (binary classification), binding affinities (regression), and mechanisms of action (activation/inhibition classification). The module employs an automated machine learning framework with multi-layer stacking and bagging techniques to optimize predictive performance across all tasks [51].
The following diagram illustrates DTIAM's integrated architecture:
Performance Benchmarking and Experimental Validation
In comprehensive benchmarking studies, DTIAM demonstrated substantial performance improvements over state-of-the-art baseline methods across all prediction tasks, particularly in challenging cold-start scenarios [51]. The framework was evaluated under three experimentally relevant settings:
- Warm Start: Both drugs and targets appear in the training data
- Drug Cold Start: Novel drugs not present in training data
- Target Cold Start: Novel targets not present in training data
DTIAM achieved superior performance in all scenarios, with particularly notable advantages in cold-start conditions where it outperformed methods including CPIGNN, TransformerCPI, MPNNCNN, and KGE_NFM on benchmark datasets [51].
For experimental validation, researchers applied DTIAM to screen a high-throughput molecular library of approximately 10 million compounds for potential TMEM16A inhibitors. TMEM16A, a calcium-activated chloride channel, represents a promising therapeutic target for various conditions including hypertension, asthma, and cancer. Following computational prediction, top-ranked candidates underwent whole-cell patch clamp experiments, which confirmed multiple effective TMEM16A inhibitors with nanomolar potency, validating DTIAM's predictive accuracy and translational potential [51].
The ability to distinguish between activation and inhibition mechanisms represents a particular advancement of the DTIAM framework. Whereas most computational methods treat drug-target interactions as simple binary events, DTIAM's MoA prediction capability provides critical functional information for drug discovery. For instance, distinguishing whether a compound activates or inhibits dopamine receptors is clinically essential, as activators may treat Parkinson's disease while inhibitors could address psychotic disorders [51].
Performance Benchmarking: Comparative Analysis of Prediction Methods
Rigorous performance assessment is essential for evaluating the relative strengths and limitations of different drug-target prediction approaches. A systematic comparison of seven target prediction methods conducted in 2025 provides comprehensive benchmarking data on the accuracy, reliability, and operational characteristics of both ligand-based and target-based methodologies [6].
Experimental Design and Evaluation Metrics
The comparative analysis employed a shared benchmark dataset of FDA-approved drugs to ensure fair and consistent evaluation across all methods [6]. The experimental protocol included:
- Dataset Preparation: Researchers compiled a benchmark set of 100 randomly selected FDA-approved drugs from ChEMBL version 34, which contains 2,431,025 compounds, 15,598 targets, and 20,772,701 interactions. To prevent evaluation bias, these molecules were excluded from the main database used for predictions [6].
- Method Selection: The study evaluated seven computational methods representing both ligand-based and target-based approaches, including stand-alone codes and web servers: MolTarPred, PPB2, RF-QSAR, TargetNet, ChEMBL, CMTNN, and SuperPred [6].
- Evaluation Framework: Performance was assessed using standard classification metrics including recall (sensitivity), precision, and overall accuracy in predicting known drug-target interactions. The study also examined practical considerations including operational requirements, database dependencies, and algorithmic characteristics [6].
Table 2: Performance Benchmarking of Drug-Target Prediction Methods
| Method | Approach Type | Algorithm | Database | Key Strengths | Performance Notes |
|---|---|---|---|---|---|
| MolTarPred | Ligand-based | 2D similarity | ChEMBL 20 | Highest effectiveness in benchmark | Most effective method in study [6] |
| PPB2 | Ligand-based | Nearest neighbor/Naïve Bayes/DNN | ChEMBL 22 | Multiple algorithm options | Moderate performance [6] |
| RF-QSAR | Target-based | Random forest | ChEMBL 20&21 | Target-specific models | Performance varies by target [6] |
| TargetNet | Target-based | Naïve Bayes | BindingDB | Multiple fingerprint types | Unclear top similar ligand [6] |
| ChEMBL | Target-based | Random forest | ChEMBL 24 | Recent database version | Morgan fingerprints [6] |
| CMTNN | Target-based | ONNX runtime | ChEMBL 34 | Latest ChEMBL data | Stand-alone code [6] |
| SuperPred | Ligand-based | 2D/fragment/3D similarity | ChEMBL & BindingDB | Multiple similarity types | Unclear top similar ligand [6] |
Optimization Strategies and Performance Trade-offs
The benchmarking study revealed several critical optimization strategies that impact method performance:
Fingerprint and Metric Selection: For ligand-based methods, the choice of molecular fingerprint and similarity metric significantly influences prediction accuracy. Specifically, Morgan fingerprints with Tanimoto scores demonstrated superior performance compared to MACCS fingerprints with Dice scores in the MolTarPred algorithm [6].
Database Quality and Filtering: Implementing high-confidence filtering of interaction data (e.g., using only interactions with confidence scores ≥7 in ChEMBL) improves prediction precision but reduces recall, making this strategy less ideal for comprehensive drug repurposing applications where maximizing potential hit identification is prioritized [6].
Recall-Precision Trade-offs: The study observed inherent trade-offs between recall (ability to identify all true interactions) and precision (accuracy of predicted interactions). Methods optimized for high recall typically generate more potential repurposing candidates but require more extensive experimental validation, while high-precision methods yield fewer candidates but with higher validation rates [6].
The comprehensive analysis concluded that MolTarPred was the most effective method overall in the benchmark, highlighting the continued competitive performance of ligand-based approaches, particularly when optimized with appropriate fingerprints and similarity metrics [6]. However, the optimal method selection depends on specific research objectives, with target-based approaches offering advantages for novel target space exploration and mechanistic insights when sufficient structural or sequence data is available [6] [51].
Successful implementation of drug repurposing and multi-target discovery research requires access to comprehensive biological databases, specialized software tools, and experimental reagents. The following table catalogs essential resources referenced in the case studies and methodological discussions, providing researchers with a practical starting point for establishing their research workflows.
Table 3: Essential Research Resources for Drug Repurposing and Multi-Target Discovery
| Resource Category | Specific Examples | Key Applications | Access Information |
|---|---|---|---|
| Bioactivity Databases | ChEMBL, BindingDB, PubChem BioAssay | Source of annotated drug-target interactions; reference data for similarity searching | Publicly available [6] [11] |
| Drug-Target Interaction Resources | DrugBank, PharmGKB, Therapeutic Target Database (TTD) | Drug mechanism information; target-disease associations; clinical trial data | Publicly available [47] [52] |
| Protein Structure Resources | Protein Data Bank (PDB), AlphaFold Protein Structure Database | Source of 3D protein structures for docking and structure-based design | Publicly available [13] [52] |
| Ligand-Based Prediction Tools | MolTarPred, SuperPred, PPB2 (Polypharmacology Browser 2) | 2D/3D similarity searching; target prediction based on chemical similarity | Stand-alone codes and web servers [6] |
| Target-Based Prediction Tools | RF-QSAR, TargetNet, CMTNN, DTIAM | Machine learning-based target prediction; binding affinity estimation | Mixed stand-alone and web-based [6] [51] |
| Molecular Docking Software | AutoDock, GOLD, Glide (Schrödinger) | Structure-based virtual screening; binding pose prediction | Commercial and academic licenses [13] [11] [52] |
| Molecular Dynamics Platforms | GROMACS, AMBER, Desmond | Simulation of drug-target interactions; binding stability assessment | Commercial and academic licenses [52] [36] |
The selection of appropriate resources should be guided by specific research objectives and experimental constraints. For rapid repurposing screening of large compound libraries, ligand-based methods with access to comprehensive bioactivity databases typically offer the most efficient approach. For target-centric discovery or mechanism of action studies, structure-based tools and advanced frameworks like DTIAM that incorporate protein information provide deeper mechanistic insights [6] [51]. The increasing availability of integrated platforms that combine multiple methodologies offers promising opportunities for comprehensive drug-target profiling that leverages the complementary strengths of both ligand-based and target-based paradigms.
The case studies and performance analyses presented in this article demonstrate that both ligand-based and target-based chemogenomic approaches provide valuable, complementary methodologies for advancing drug repurposing and multi-target discovery. Ligand-based methods like MolTarPred offer computational efficiency and strong performance when reference ligand data is available, while target-based approaches including the DTIAM framework enable novel target exploration and mechanistic insights, particularly through the integration of self-supervised learning and multi-task prediction [6] [51].
The future landscape of drug repurposing and polypharmacology research will likely be shaped by several emerging trends. The integration of artificial intelligence and machine learning continues to advance predictive accuracy, with self-supervised learning frameworks addressing the critical challenge of limited labeled data [49] [51]. Additionally, the growing availability of high-quality protein structures through experimental methods and AlphaFold predictions is expanding the applicability of structure-based approaches [6] [13]. Furthermore, the systematic incorporation of multi-omics data and heterogeneous biological networks is enabling more comprehensive modeling of drug effects across multiple biological scales [49] [51] [48].
Despite these advancements, significant challenges remain in achieving optimal integration of computational predictions with experimental validation. The translation of in silico hits to clinically effective repurposed drugs requires careful consideration of therapeutic dosing, patient selection strategies, and intellectual property landscapes [47] [48]. The promising results from successful repurposing cases—from mebendazole's anticancer applications to DTIAM's novel TMEM16A inhibitors—provide compelling evidence that systematic computational approaches can unlock substantial hidden therapeutic potential in existing drugs [6] [51] [48]. As these methodologies continue to mature, they will increasingly enable researchers to navigate the complex landscape of polypharmacology and accelerate the discovery of new therapeutic applications for existing drugs, ultimately expanding treatment options for patients across diverse disease areas.
Navigating Challenges and Enhancing Predictive Performance
Addressing Data Sparsity, the 'Cold Start' Problem, and Class Imbalance
The systematic prediction of drug-target interactions (DTIs) is a cornerstone of modern drug discovery, enabling the identification of novel therapeutic agents, drug repurposing, and the understanding of polypharmacology [43] [11]. Chemogenomic approaches, which integrate chemical and genomic information, have emerged as powerful computational tools for this task. These methods are broadly categorized into ligand-based (predicting targets based on known ligands), target-based (predicting ligands based on target structures or sequences), and hybrid methods that combine both philosophies [6] [10]. However, the development and application of these models are consistently hampered by three interconnected data challenges: data sparsity, where the known interaction matrix is overwhelmingly incomplete; the 'cold start' problem, which refers to the inability to make predictions for new drugs or targets that have no known interactions; and class imbalance, where the number of known positive interactions is vastly outnumbered by unknown or negative pairs [53] [13] [43]. This guide objectively compares the performance of contemporary ligand-based and target-based methods in overcoming these hurdles, providing researchers with a clear view of the current methodological landscape.
Performance Comparison of Ligand-Based and Target-Based Methods
The following tables summarize a systematic comparison of various target prediction methods, highlighting their performance and inherent limitations regarding the core challenges. The data is synthesized from a 2025 benchmark study that evaluated methods using a shared dataset of FDA-approved drugs [6].
Table 1: Overview and Comparative Performance of Target Prediction Methods
| Method | Type | Core Algorithm | Key Strength | Inherent Challenge |
|---|---|---|---|---|
| MolTarPred [6] | Ligand-based | 2D Similarity (Morgan Fingerprints) | High overall accuracy & interpretability | Cold-start (new drugs) |
| PPB2 [6] | Ligand-based | Nearest Neighbor/Naïve Bayes/DNN | Integrates multiple algorithms | Cold-start (new drugs) |
| SuperPred [6] | Ligand-based | 2D/Fragment/3D Similarity | Multi-faceted similarity search | Cold-start (new drugs) |
| RF-QSAR [6] | Target-centric | Random Forest (QSAR) | Good for novel protein targets | Relies on bioactivity data for training |
| TargetNet [6] | Target-centric | Naïve Bayes | Utilizes multiple fingerprint types | Relies on bioactivity data for training |
| ChEMBL [6] | Target-centric | Random Forest (Morgan FP) | Extensive, validated bioactivity data | Relies on bioactivity data for training |
| CMTNN [6] | Target-centric | Multitask Neural Network | High performance on trained targets | Cold-start (new targets), low interpretability |
| KGE_NFM [54] | Hybrid (KG) | Knowledge Graph Embedding + NFM | Excels in cold-start for proteins | Complex framework, requires diverse data |
| MGDTI [53] | Hybrid (Graph) | Meta-learning + Graph Transformer | Specifically designed for cold-start | Computationally intensive |
Table 2: Quantitative Benchmarking on a Shared Dataset (100 FDA-Approved Drugs) [6]
| Method | Recall | Impact of High-Confidence Filtering | Optimal Configuration |
|---|---|---|---|
| MolTarPred | Most effective | Recall decreases (less ideal for repurposing) | Morgan fingerprints + Tanimoto score |
| Ligand-based (General) | Varies with similarity threshold | Reduces false positives but increases sparsity | Dependent on fingerprint & similarity metric |
| Target-centric (General) | Dependent on training data coverage | Improves precision but can exacerbate cold-start | N/A |
| KGE_NFM [54] | High AUPR (0.961 on balanced data) | Robust performance under data imbalance | Combination of KG and recommendation system |
| MGDTI [53] | Superior in cold-start scenarios | Utilizes similarity to mitigate interaction scarcity | Meta-learning adaptation for cold-drug/cold-target tasks |
Experimental Protocols for Benchmarking DTI Methods
To ensure fair and reproducible comparisons, rigorous experimental protocols are essential. The following methodology is adapted from recent large-scale benchmark studies [6] [54].
Database Curation and Preparation
- Data Source: The ChEMBL database (version 34) is typically used for its extensive and experimentally validated bioactivity data, containing over 2.4 million compounds and 15,000 targets [6].
- Interaction Data Extraction: Bioactivity records with standard values (IC₅₀, Kᵢ, or EC₅₀) below 10,000 nM are retrieved. Entries associated with non-specific or multi-protein targets are filtered out, and duplicate compound-target pairs are removed to ensure data quality [6].
- Confidence Scoring: A high-confidence dataset is often created by filtering interactions with a minimum confidence score of 7 (indicating a direct protein complex subunit assignment) to reduce noise [6].
- Benchmark Dataset: A benchmark dataset is prepared from FDA-approved drugs. To prevent bias, these molecules are excluded from the main database used for model training or similarity searching, ensuring the evaluation simulates the prediction of novel interactions [6].
Evaluation Setup and Metrics
- Prediction Tasks: Methods are tasked to predict targets for query molecules in the benchmark dataset.
- Performance Metrics: Recall (the proportion of actual positives correctly identified) is a critical metric, especially for drug repurposing where finding all potential targets is key. The impact of high-confidence filtering on recall is a key analysis point [6]. For classification tasks, Area Under the Precision-Recall Curve (AUPR) is used, particularly under imbalanced data conditions [54].
- Cold-Start Evaluation: Models are specifically tested under two scenarios [53]:
- Cold-Drug: Predicting interactions for new drugs with no known targets.
- Cold-Target: Predicting interactions for new targets with no known ligands.
Diagram 1: Experimental workflow for DTI method benchmarking.
Successful DTI prediction research relies on a suite of computational tools and databases. The table below lists key resources used in the featured experiments and the broader field [6] [13] [55].
Table 3: Key Research Reagent Solutions for DTI Prediction
| Resource Name | Type | Primary Function | Application in Experiments |
|---|---|---|---|
| ChEMBL [6] | Database | Repository of bioactive molecules, targets, and interactions | Primary source for curated bioactivity data and ligand-target pairs. |
| RDKit [55] | Software Library | Cheminformatics and machine learning | Processing SMILES strings, generating molecular fingerprints (e.g., Morgan). |
| PostgreSQL / pgAdmin4 [6] | Database Tool | Management of relational databases | Hosting and querying local instances of the ChEMBL database. |
| Morgan Fingerprints [6] | Molecular Descriptor | Representation of molecular structure | Used as input for similarity calculations (MolTarPred) and ML models (RF-QSAR). |
| SMILES [55] | Molecular Representation | String-based notation of chemical structures | Standardized representation of query drugs and database molecules. |
| Knowledge Graphs (e.g., PharmKG) [54] [13] | Data Framework | Integrating heterogeneous biological data | Providing multi-modal context to overcome data sparsity and cold-start. |
| AlphaFold [13] | Protein Structure Tool | Predicting 3D protein structures | Generating structural data for target-based methods when experimental structures are unavailable. |
Analysis of Results and Strategic Recommendations
The comparative data reveals a clear trade-off between ligand-based and target-based approaches, heavily influenced by the specific data challenge at hand.
- Combating Data Sparsity: Hybrid models that integrate multiple data sources show the most promise. KGE_NFM, which learns low-dimensional representations from knowledge graphs, achieves robust performance (AUPR = 0.961) even when the interaction matrix is sparse and noisy [54]. This approach does not rely solely on the DTI matrix but enriches it with heterogeneous data, effectively filling in gaps.
- Solving the Cold-Start Problem: This remains a key weakness for pure ligand-based and target-centric methods, which require known ligands or bioactivity data, respectively [6] [43]. MGDTI, a graph-based model trained with meta-learning, is specifically designed for this scenario and demonstrates superior performance by rapidly adapting to new drugs or targets with limited interaction data [53]. For cold-start scenarios involving new proteins, KGE_NFM also shows outstanding results [54].
- Mitigating Class Imbalance: High-confidence filtering improves precision but reduces recall, which can be detrimental for drug repurposing tasks where finding all potential targets is the goal [6]. Model-level solutions are more effective. KGE_NFM and other heterogeneous data methods maintain stable AUPR scores even when the ratio between positive and negative samples is unbalanced, unlike traditional feature-based methods whose performance can drop by over 10% [54].
Diagram 2: Strategic solution mapping for core DTI challenges.
The landscape of DTI prediction is evolving from pure ligand-based or target-based models toward sophisticated hybrid frameworks. For researchers, the choice of method should be dictated by the specific problem:
- For well-established targets with ample bioactivity data, target-centric methods like CMTNN or ligand-based similarity searches like MolTarPred offer strong, interpretable predictions.
- For novel target discovery or predicting effects for new drugs, hybrid models like KGE_NFM and MGDTI that are designed to handle data sparsity and the cold-start problem are unequivocally superior.
The integration of knowledge graphs, meta-learning, and advanced feature engineering using tools like AlphaFold and large language models represents the future direction for building more accurate, robust, and generalizable DTI prediction systems [54] [53] [13].
The pursuit of novel therapeutic compounds increasingly relies on two distinct computational philosophies: ligand-based drug design (LBDD) and structure-based drug design (SBDD). LBDD utilizes information from known active ligands to predict new candidates, while SBDD directly leverages the three-dimensional structure of the target protein. A particularly advanced concept within both approaches is ligand bias—the ability of a ligand to preferentially activate specific downstream signaling pathways of a receptor, most prominently G protein-coupled receptors (GPCRs), over others. This paradigm promises therapeutics with maximized efficacy and minimized on-target side-effects [56]. This guide provides an objective comparison of these strategies, focusing on their interplay with ligand bias and their respective dependencies on 3D structural data. We present supporting experimental data, detailed methodologies, and key resources to equip researchers with the tools needed to navigate these complementary approaches.
Core Concepts and Comparative Framework
Ligand-Based Drug Design (LBDD)
LBDD operates on the principle of "molecular similarity," where compounds structurally similar to known active ligands are likely to exhibit similar biological activity. Key techniques include Quantitative Structure-Activity Relationship (QSAR) modeling, which builds mathematical models linking chemical features to biological activity, and pharmacophore modeling, which identifies the essential steric and electronic features responsible for a ligand's interaction with the target [57]. A significant advantage of LBDD is its applicability when the three-dimensional structure of the target protein is unknown or difficult to obtain. It allows for the rapid virtual screening of large compound libraries at a relatively low computational cost [58] [57].
Structure-Based Drug Design (SBDD)
SBDD requires the three-dimensional structure of the target protein, obtained through methods like X-ray crystallography, NMR, or cryo-electron microscopy (cryo-EM). The primary technique is molecular docking, which predicts how a small molecule (ligand) binds to a protein's binding pocket and scores the stability of that interaction [58] [57]. This approach allows for the direct visualization of interaction sites and facilitates the rational design of molecules with high affinity and specificity. However, its major limitation is the dependency on a high-quality protein structure and the challenges associated with accounting for full protein flexibility and accurate binding affinity prediction [58] [57].
The Emergence of Ligand Bias in GPCR Drug Discovery
Ligand bias, or functional selectivity, describes the phenomenon where a ligand stabilizes a specific active receptor conformation, leading to preferential activation of one signaling pathway (e.g., G protein) over another (e.g., β-arrestin) [59] [56]. This offers a powerful mechanism to separate therapeutic effects from adverse side-effects. For instance, G protein-biased μ-opioid receptor (MOR) agonists are being developed as analgesics with reduced respiratory depression and gastrointestinal issues, which are traditionally linked to the β-arrestin pathway [59] [56]. The accurate quantification of bias is therefore critical, with the operational model of agonism and the simplified Δlog(Emax/EC50) method being gold standards for calculating pathway bias from in vitro signaling data [59].
Quantitative Comparison of Approaches and Outcomes
The table below summarizes key performance metrics and characteristics of LBDD and SBDD, particularly in the context of identifying biased ligands.
Table 1: Quantitative and Qualitative Comparison of LBDD and SBDD
| Aspect | Ligand-Based Design (LBDD) | Structure-Based Design (SBDD) |
|---|---|---|
| Primary Data Input | Structures & activities of known ligands [57] | 3D structure of the target protein [57] |
| Key Computational Techniques | QSAR, Pharmacophore Modeling, 2D/3D Similarity Search [57] | Molecular Docking, Molecular Dynamics (MD) Simulations [58] [57] |
| Bias Assessment Method | Inference from ligand structure-activity relationships (SAR) | Analysis of ligand-induced receptor conformations via MD and machine learning [60] |
| Typical Throughput | High (suitable for HTS of large libraries) [59] | Medium to Low (more computationally demanding) [58] |
| Success Metric (Bias Identification) | Identification of novel chemotypes with predicted bias from ligand data [58] | De novo design of ligands that stabilize specific receptor states [61] [60] |
| Key Limitations | Bias is towards the chemical space of known templates; cannot explain structural mechanism of bias [58] | Dependent on availability and quality of protein structures; struggles with predicting functional efficacy [62] [57] |
| Reported Hit Rate (Examples) | Hit rates competitive with HTS; specific numbers target-dependent [58] | Identification of nM inhibitors (e.g., HDAC8 IC50 2.7 nM) via hybrid workflows [58] |
Table 2: Experimental Outcomes in Biased Ligand Development
| Target / Therapeutic Area | Approach | Key Outcome | Reference / Compound |
|---|---|---|---|
| μ-Opioid Receptor (Pain) | HTS & Bias Quantification (Δlog(Emax/EC50)) | Identified 440 hits with >10-fold Gαi bias vs. reference agonist DAMGO [59] | [59] |
| Type 1 Parathyroid Hormone Receptor (PTHR1) | Nanobody-Tethered Ligand (SBDD) | Created PTH1-11-Nb, the most Gαs/cAMP-biased PTHR1 agonist known [63] | [63] |
| Angiotensin II Type 1 Receptor (Heart Failure) | Rational Design of Biased Ligand | TRV027: β-arrestin-biased agonist; inhibited Gαq-mediated vasoconstriction [56] | TRV027 [56] |
| Kinases (Oncology) | Machine Learning on Sequence & Affinity Data | Models predict binding affinity from protein sequence & ligand SMILES, but performance drops with proper, unbiased data splits [62] | Davis & KIBA Data Sets [62] |
Detailed Experimental Protocols for Biased Ligand Research
Protocol 1: High-Throughput Quantification of Ligand Bias
This protocol, adapted from an industrial-scale screening campaign for biased μ-opioid receptor agonists, details the steps for quantifying ligand bias across two pathways [59].
- Cell Line Engineering: Generate stable cell lines expressing the target GPCR. For instance, use CHO-K1 cells for G protein signaling and U2OS cells engineered with a β-arrestin enzyme fragment complementation assay (e.g., PathHunter cells) [59].
- Assay Development: Establish homogenous, robust signaling assays for each pathway.
- G Protein Signaling: Implement a cAMP assay or GTPγS binding for Gαi-coupled receptors, ensuring a wide dynamic range.
- β-arrestin Recruitment: Use a commercial β-arrestin recruitment assay (e.g., DiscoverRx PathHunter) [59].
- Compound Screening: In a 384-well format, test a diverse compound library with 11-point, half-log concentration-response curves. Include a reference unbiased agonist (e.g., DAMGO for MOR) in every experiment for normalization [59].
- Data Analysis and Bias Calculation:
- For each compound and the reference agonist, determine the EC50 and Emax for both pathways.
- Calculate the Δlog(Emax/EC50) value for each pathway relative to the reference agonist.
- The bias between two pathways (e.g., G protein vs. β-arrestin) is given by ΔΔlog(Emax/EC50) = Δlog(Emax/EC50)Pathway A - Δlog(Emax/EC50)Pathway B [59].
- Validate a subset of hits using the more rigorous operational model to confirm the bias calculated by the simplified method.
Protocol 2: An Interaction-Aware 3D Molecular Generative Framework
This protocol describes a structure-based, deep learning approach for designing ligands tailored to specific protein-ligand interactions, thereby implicitly guiding bias [61].
- Data Curation: Curate a set of high-quality protein-ligand complex structures from a database like PDBbind [61] [62].
- Interaction Condition Setting: For a given target binding pocket, define an "interaction condition."
- During Training: Use software like the Protein-Ligand Interaction Profiler (PLIP) to analyze crystallographic complexes and extract interaction patterns (hydrogen bonds, salt bridges, hydrophobic, π-π stacking) for the native ligand [61].
- During Generation (Reference-Free): Predefine chemical rules to classify protein atoms into roles (e.g., hydrogen bond donor/acceptor, cationic, anionic, aromatic) without a reference ligand [61].
- Model Training: Train a deep generative model (e.g., DeepICL) to sequentially add atoms to a growing ligand. The model is conditioned on:
- The 3D coordinates of the protein pocket atoms.
- The local interaction condition (a one-hot vector indicating the interaction type for nearby protein atoms) [61].
- Ligand Generation and Evaluation:
- De Novo Design: Start from a manually selected seed point inside the pocket and generate complete ligands.
- Ligand Elaboration: Start from an existing core structure (e.g., a fragment) and elaborate it.
- Assess generated ligands for binding pose stability, affinity (via docking scores), and similarity to the desired interaction pattern [61].
Visualization of Workflows and Signaling
The following diagrams illustrate the core experimental workflow for bias quantification and the conceptual signaling pathways involved in ligand bias.
Diagram Title: Bias Quantification Workflow
Diagram Title: Biased Signaling at a GPCR
The Scientist's Toolkit: Essential Research Reagents and Solutions
Table 3: Key Research Reagents for Biased Ligand Studies
| Reagent / Tool | Function / Application | Example Product / Source |
|---|---|---|
| PathHunter β-arrestin Assay | Measures β-arrestin recruitment to GPCRs in a high-throughput, homogenous format. | DiscoverRx (Now part of Eurofins) [59] |
| Cryo-EM Services | Determines high-resolution 3D structures of membrane proteins (e.g., GPCRs) in different states. | Commercial structural biology services [57] |
| PDBbind Database | Curated database of protein-ligand complex structures and binding affinities for model training and validation. | PDBbind [61] [62] |
| GPCR Stable Cell Lines | Engineered cell lines (e.g., CHO, HEK293) overexpressing specific GPCRs for consistent signaling assays. | Commercial vendors (e.g., DiscoverRx, Thermo Fisher) [59] [63] |
| PLIP (Protein-Ligand Interaction Profiler) | Open-source tool for automated detection of non-covalent interactions in PDB structures. | https://plip-tool.biotec.tu-dresden.de [61] |
| Molecular Docking Software (AutoDock Vina) | Widely used program for predicting ligand binding poses and scoring affinity. | Open-Source [62] |
| Nanobodies (VHH) | Small, stable antibody fragments used as conformational sensors or tethering devices for biased ligands. | Recombinant expression & selection [63] |
The dichotomy between ligand bias and dependency on 3D structures is becoming increasingly blurred with the advent of integrated and hybrid methodologies. LBDD offers speed and applicability when structural data is scarce, but it risks chemical space stagnation and cannot directly illuminate the structural mechanisms underpinning bias [58]. Conversely, SBDD provides a rational, structure-guided path to design and can exploit atomic-level details to understand bias, but it is gated by the significant challenge of obtaining relevant, high-quality structures [57].
The most promising future lies in hybrid strategies that leverage the strengths of both. For example, ligand-based pharmacophore models can pre-filter compound libraries, which are then subjected to more computationally intensive structure-based docking and molecular dynamics simulations [58]. Furthermore, breakthroughs like nanobody tethering demonstrate how structural insights can be used to engineer extreme bias not achievable by modifying the ligand's core structure alone [63]. Meanwhile, machine learning models trained on both ligand chemical data and protein structural/sequence information are poised to revolutionize the field, provided that biases in training data (like sequence or ligand similarity) are carefully managed to ensure generalizability [62] [60].
In conclusion, the objective comparison reveals that neither the ligand bias paradigm nor the 3D structure-dependent approach is universally superior. The choice of strategy depends on the specific target, the available data, and the stage of the drug discovery campaign. A deliberate, integrated approach that combines the scalability of ligand-based methods with the mechanistic insight of structure-based design offers the most robust path forward for discovering the next generation of safer, more effective biased therapeutics.
In modern computational drug discovery, the choice between ligand-based and target-based chemogenomic approaches is fundamental. Ligand-based methods predict interactions by comparing chemical similarity to known active compounds, while target-based approaches use protein structure or sequence information to model binding events [6] [43]. The performance of both paradigms heavily depends on implementing effective optimization strategies to enhance predictive accuracy, reduce false positives, and ensure computational efficiency. This guide objectively compares three critical optimization classes—feature selection, ensemble learning, and high-confidence filtering—by analyzing their implementation across methodological frameworks and evaluating their impact on key performance metrics using recent experimental data.
The following sections provide a detailed comparison of these strategies, summarizing quantitative performance data, detailing experimental protocols, and illustrating methodological workflows. These analyses provide researchers with a evidence-based framework for selecting and implementing optimization strategies that best address specific drug discovery challenges.
Performance Comparison of Optimization Strategies
Experimental data from systematic evaluations demonstrate how different optimization strategies impact predictive performance across various computational methods.
Table 1: Performance Metrics of Target Prediction Methods Utilizing Different Optimization Strategies
| Method | Optimization Strategy | Key Implementation Details | Performance Impact | Primary Use Case |
|---|---|---|---|---|
| MolTarPred [6] | High-confidence Filtering | Confidence score ≥7 (ChEMBL); Morgan fingerprints [6] | Highest overall effectiveness; Recall reduction with high-confidence filter [6] | Ligand-based target fishing |
| EnsemKRR [64] | Ensemble Learning + Dimensionality Reduction | Kernel Ridge Regression base learners; Feature subspacing [64] | AUC: 94.3% (Highest among compared methods) [64] | General DTI prediction |
| EnsemDT [64] | Ensemble Learning + Dimensionality Reduction | Decision Tree base learners; Multiple DR techniques [64] | Outperforms SVM/RF; Improved with dimensionality reduction [64] | General DTI prediction |
| RF-QSAR [6] | Ensemble Learning | Random Forest algorithm; ECFP4 fingerprints [6] | Benchmarkable performance (Precise metrics not fully reported) [6] | Target-centric prediction |
| Feature Selection + RF [65] | Feature Selection + Ensemble Learning | Correlation, IG, Chi-Square, Relief; Random Forest classifier [65] | Improved accuracy with selected feature subsets [65] | Handling high-dimensional data |
| CMTNN [6] | Algorithmic Optimization | Multitask Neural Network; ONNX runtime [6] | Benchmarkable performance (Precise metrics not fully reported) [6] | Target-centric prediction |
Table 2: Impact of Data Filtering and Feature Choices on Model Performance
| Optimization Parameter | Options Compared | Performance Outcome | Interpretation |
|---|---|---|---|
| Fingerprint Type [6] | Morgan (ECFP-like) vs. MACCS | Morgan fingerprints with Tanimoto outperformed MACCS with Dice [6] | Morgan fingerprints capture richer structural features |
| Confidence Filtering [6] | ChEMBL confidence score ≥7 vs. lower thresholds | Increased precision but reduced recall [6] | Trade-off between data quality and coverage |
| Data Balancing [65] | ROS, SMOTE, Adaptive SMOTE | Addressing imbalance improved overall model accuracy [65] | Critical for realistic DTI prediction where negatives dominate |
| Similarity Metric [6] | Tanimoto vs. Dice | Tanimoto with Morgan fingerprints provided superior accuracy [6] | Metric choice interacts with fingerprint representation |
Experimental Protocols and Methodologies
High-Confidence Filtering Implementation
High-confidence filtering ensures model training uses only highly reliable interaction data. A representative protocol from a recent systematic comparison involves [6]:
- Database Sourcing: Retrieve bioactivity data from established databases like ChEMBL (version 34 used in the study), which contains experimentally validated drug-target interactions, inhibitory concentrations, and binding affinities [6].
- Activity Thresholding: Filter records to include only those with standard values (IC₅₀, Kᵢ, or EC₅₀) below 10,000 nM, ensuring retained interactions have meaningful potency [6].
- Complexity Reduction: Remove entries associated with non-specific or multi-protein targets by filtering out target names containing keywords like "multiple" or "complex." Eliminate duplicate compound-target pairs [6].
- Confidence Scoring: Apply a confidence score filter. For example, in ChEMBL, a confidence score of 7 indicates "direct protein complex subunits assigned." Retain only interactions meeting this high-confidence threshold [6].
- Benchmark Preparation: To prevent bias, remove molecules intended for benchmarking (e.g., a set of FDA-approved drugs) from the main database before model training [6].
Ensemble Learning with Dimensionality Reduction
This protocol combines multiple learners with feature reduction to enhance prediction of Drug-Target Interactions (DTI) [64]:
- Feature Subspacing: Randomly divide the complete feature set (e.g., drug descriptors and target sequence descriptors) into multiple, smaller subspaces. This injects diversity into the ensemble [64].
- Dimensionality Reduction (DR): Apply a DR technique like Singular Value Decomposition (SVD), Partial Least Squares (PLS), or Laplacian Eigenmaps to each feature subspace. This reduces noise and computational cost [64].
- Base Learner Training: Train homogeneous base learners (e.g., Decision Trees or Kernel Ridge Regression models) on each processed, reduced feature subspace [64].
- Prediction Aggregation: For a new drug-target pair, generate predictions from all base learners. Compute the final prediction by averaging the scores (for regression) or using majority voting (for classification) [64].
Feature Selection for Drug-Target Interaction Prediction
This methodology identifies the most informative features to improve model interpretability and performance [65]:
- Data Acquisition and Preprocessing: Source protein data from KEGG and drug data from DrugBank. Represent drugs via fingerprints or descriptors and proteins via sequence-based features [65].
- Handling Class Imbalance: Address the inherent imbalance in DTI data (few known interactions vs. many non-interactions) using techniques like Random Over-Sampling (ROS), Synthetic Minority Over-sampling Technique (SMOTE), or Adaptive SMOTE [65].
- Feature Selection Method Application: Evaluate multiple feature selection methods on the balanced dataset. Common methods include:
- Correlation-based: Removes highly correlated redundant features.
- Information Gain (IG): Ranks features by their mutual information with the target variable.
- Chi-Square (CS): Tests independence of features from the target class.
- Relief: Estimates feature quality based on how well their values distinguish between nearby instances [65].
- Model Training and Validation: Train classifiers (e.g., Support Vector Machine, Random Forest, AdaBoost, Logistic Regression) using the selected feature subsets. Validate performance via cross-validation, using metrics like AUC-ROC [65].
High-Confidence Data Filtering Workflow
Ensemble Learning with Dimensionality Reduction
Successful implementation of the optimization strategies discussed requires leveraging specific computational tools and data resources.
Table 3: Key Research Reagents and Computational Resources
| Resource Name | Type | Primary Function in Optimization | Application Context |
|---|---|---|---|
| ChEMBL Database [6] | Bioactivity Database | Source of experimentally validated interactions for high-confidence filtering and model training. | Ligand-based and target-centric methods |
| Molecular Fingerprints (Morgan/ECFP) [6] | Molecular Representation | Captures chemical structure; Choice significantly impacts ligand-based prediction accuracy. | Ligand-based target fishing, similarity search |
| RDKit | Cheminformatics Toolkit | Generates molecular fingerprints and descriptors; Facilitates feature extraction for machine learning. | General pre-processing and feature engineering |
| Random Forest | Machine Learning Algorithm | Ensemble method that aggregates predictions from multiple decision trees, reducing overfitting. | General classification and regression for DTI |
| SMOTE [65] | Data Balancing Algorithm | Generates synthetic minority class samples to handle imbalanced drug-target interaction datasets. | Pre-processing for model training |
| Feature Selection Algorithms [65] | Pre-processing Module | Identifies most predictive features from high-dimensional drug and target data (e.g., IG, Chi-Square). | Handling high-dimensional feature spaces |
The strategic implementation of feature selection, ensemble learning, and high-confidence filtering is paramount for optimizing chemogenomic drug discovery pipelines. Empirical comparisons reveal that high-confidence filtering prioritizes precision at the cost of recall, making it ideal for lead optimization, while ensemble methods like EnsemKRR achieve top-tier overall predictive accuracy (AUC 94.3%) for broad interaction screening [6] [64]. The optimal strategy is context-dependent, dictated by project goals, data availability, and the fundamental choice between ligand-based and target-based approaches. By leveraging the protocols, performance data, and resources outlined in this guide, researchers can make informed decisions to enhance the efficiency and success rate of their computational drug discovery efforts.
The accurate prediction of interactions between small molecules and their protein targets is a fundamental challenge in modern drug discovery. Traditional computational approaches have largely fallen into two distinct categories: ligand-based (LB) and structure-based (SB) methods. Ligand-based methods, rooted in the principle that similar molecules tend to have similar biological activities, predict targets by comparing a query compound to a database of known active ligands [43] [11]. In contrast, structure-based methods, such as molecular docking, rely on the three-dimensional (3D) structure of a target protein to simulate how a ligand might bind, estimating interaction likelihood based on binding affinity and complementarity [6] [64]. While LB methods are powerful when abundant ligand data exists, their performance degrades when similar ligands are scarce. SB methods provide a mechanistic view of binding but are constrained by the availability of high-quality protein structures and can be computationally intensive [43] [11].
The integration of these approaches into hybrid and sequential workflows represents a paradigm shift, moving beyond the limitations of individual methods to achieve superior predictive performance. This guide objectively compares the performance of standalone and integrated methods, providing experimental data and detailed protocols to guide researchers in implementing these advanced strategies for more efficient and reliable drug discovery and repurposing.
Performance Comparison: Standalone vs. Integrated Approaches
Systematic benchmarking is crucial for selecting the right computational tool. A 2025 study provided a precise comparison of seven target prediction methods using a shared benchmark dataset of FDA-approved drugs, offering a clear view of their relative performance [6].
Table 1: Performance Comparison of Popular Target Prediction Methods [6]
| Method | Type | Key Algorithm | Key Finding | Recall (High-Confidence Filter) |
|---|---|---|---|---|
| MolTarPred | Ligand-centric | 2D Similarity | Most effective method in benchmark | Reduced (less ideal for repurposing) |
| RF-QSAR | Target-centric | Random Forest | -- | -- |
| TargetNet | Target-centric | Naïve Bayes | -- | -- |
| ChEMBL | Target-centric | Random Forest | -- | -- |
| CMTNN | Target-centric | Neural Network | -- | -- |
| PPB2 | Ligand-centric | Nearest Neighbor/Neural Network | -- | -- |
| SuperPred | Ligand-centric | 2D/Fragment/3D Similarity | -- | -- |
The study concluded that MolTarPred was the most effective method among those evaluated [6]. Furthermore, it highlighted that model optimization strategies, such as applying high-confidence filters, can reduce recall. This trade-off makes such filtering less ideal for drug repurposing tasks, where the goal is to identify all potential targets, but may be beneficial when high-precision predictions are required [6].
The integration of LB and SB methods often yields better results than either approach alone. Another line of research focuses on chemogenomic models that inherently combine information from both ligands and targets. For instance, a 2023 study developed an ensemble chemogenomic model that integrates multi-scale information from chemical structures and protein sequences [66]. When validated on external datasets, this model demonstrated that over 45% of known targets were identified within the top-10 predictions, confirming its powerful capability to narrow down potential targets for experimental testing [66].
Table 2: Performance of an Ensemble Chemogenomic Model on External Validation [66]
| Validation Set | Targets in Top 1 | Targets in Top 10 | Enrichment Factor (Top 10) |
|---|---|---|---|
| Stratified 10-Fold CV | 26.78% | 57.96% | ~50-fold |
| External Datasets (e.g., Natural Products) | -- | >45% | -- |
Experimental Protocols for Method Benchmarking
To ensure fair and reproducible comparisons, the following protocols detail the key experimental steps for benchmarking target prediction methods.
Protocol 1: Benchmarking Setup for LB and SB Target Prediction
This protocol is based on the methodology used in the 2025 comparative study [6].
Dataset Curation:
- Source: Utilize a comprehensive, high-quality database such as ChEMBL (version 34 used in the study) [6].
- Compounds: Collect a set of FDA-approved drugs. To prevent over-optimistic performance, ensure these molecules are excluded from the main database used for prediction to avoid overlap [6].
- Interactions: Retrieve bioactivity data (e.g., IC50, Ki, EC50) with standard values below 10,000 nM. Filter for high-confidence interactions (e.g., a minimum confidence score of 7 in ChEMBL) to ensure data quality [6].
- Redundancy Removal: Remove duplicate compound-target pairs and exclude entries associated with non-specific or multi-protein targets [6].
Method Selection and Execution:
- Select a representative set of stand-alone codes and web servers, such as MolTarPred, PPB2, RF-QSAR, TargetNet, ChEMBL, CMTNN, and SuperPred [6].
- For each method, run predictions on the benchmark dataset of query molecules. For web servers, this may require manual query submission, while stand-alone codes can be run locally [6].
Performance Evaluation:
- Primary Metric: Use Recall (the proportion of actual positives that are correctly identified) to measure the ability to retrieve known interactions [6].
- Analysis: Evaluate the impact of optimization strategies, such as high-confidence filtering or the use of different molecular fingerprints (e.g., Morgan fingerprints vs. MACCS) and similarity metrics (e.g., Tanimoto vs. Dice scores) on recall [6].
Protocol 2: Implementing a Hybrid Computational-Experimental Workflow
This protocol is adapted from a 2022 study on the design of self-assembling peptides, demonstrating a powerful active learning framework for hybrid discovery [67].
Define the Molecular Design Space: Identify the family of molecules to be explored (e.g., π-conjugated peptides with oligopeptide wings up to five amino acids in length) [67].
Establish Parallel Screening Tracks:
- High-Throughput Computational Screening: Use all-atom molecular dynamics (MD) simulations to predict the structural morphology and key assembly metrics (e.g., average number of contacts per molecule, radius of gyration) for a large number of candidates [67].
- Low-Throughput Experimental Screening: Synthesize a select subset of candidates and characterize their assembly properties using techniques like UV-visible spectroscopy to measure spectral shifts indicative of desired aggregation [67].
Integrate Data via Active Learning:
- Modeling: Use deep representational learning (e.g., regularized autoencoders) to create a shared low-dimensional latent space embedding of the molecular design space. Train surrogate models (e.g., Gaussian Process Regression) on the accumulated computational and experimental data to predict the performance of untested candidates [67].
- Candidate Selection: Employ multi-objective Bayesian optimization to asynchronously select the most promising molecules for the next round of computational or experimental screening based on the model's predictions [67].
Validation: Experimentally validate the top-performing molecules identified by the workflow to confirm their predicted properties [67].
Visualizing a Sequential Hybrid Workflow
The following diagram illustrates a generalized sequential workflow that integrates computational and experimental methods, embodying the principles of the hybrid approach.
Successful implementation of hybrid workflows relies on a foundation of specific data, software, and experimental tools.
Table 3: Essential Resources for Hybrid Target Prediction Workflows
| Category | Resource Name | Function and Application |
|---|---|---|
| Bioactivity Databases | ChEMBL [6] [66] | A manually curated database of bioactive molecules with drug-like properties, providing binding affinities and target information for LB models and benchmarking. |
| DrugBank [8] [64] | A comprehensive resource containing detailed information about drugs, their mechanisms, interactions, and target data, crucial for validation. | |
| BindingDB [66] | A public database of measured binding affinities, focusing primarily on interactions between drug-like molecules and protein targets. | |
| Computational Tools | MolTarPred [6] | A ligand-centric target prediction method using 2D similarity, identified as a top-performing stand-alone tool. |
| AutoDock [11] | A widely used suite of automated docking tools, enabling SB prediction of how small molecules bind to a receptor of known 3D structure. | |
| Ensemble Models (EnsemKRR) [64] | Machine learning frameworks that combine multiple base learners and dimensionality reduction to improve DTI prediction accuracy. | |
| Experimental Assays | Binding Affinity Assays [43] | Wet-lab techniques (e.g., Ki, IC50 measurements) to quantitatively determine the strength of a drug-target interaction, serving as the ultimate validation. |
| UV-Visible Spectroscopy [67] | Used in hybrid workflows to characterize the self-assembly and aggregation behavior of molecules, providing experimental feedback for computational models. |
The integration of ligand-based and structure-based methods into hybrid and sequential workflows represents a superior strategy for drug target prediction. Quantitative benchmarks reveal that while certain standalone methods like MolTarPred excel, the combination of approaches through ensemble chemogenomic models or active learning frameworks consistently delivers more robust and reliable results. By leveraging the complementary strengths of LB and SB methods—mitigating the data scarcity issues of the former and the structural limitation of the latter—researchers can significantly narrow the candidate search space for experimental validation. The provided protocols, performance data, and toolkit offer a foundation for scientists to implement these advanced workflows, ultimately accelerating the pace of drug discovery and repurposing.
Benchmarking, Validation, and Strategic Decision-Making
The accurate prediction of drug-target interactions (DTIs) is a cornerstone of modern drug discovery, enabling the identification of new therapeutic targets, drug repurposing, and the understanding of polypharmacology [6] [13]. Computational methods for DTI prediction have largely evolved into two dominant, complementary paradigms: ligand-based and target-based (structure-based) chemogenomic approaches [10] [20].
Ligand-based methods operate on the principle that molecules with similar structural features are likely to exhibit similar biological activities and target profiles [11] [3]. These approaches are invaluable when the three-dimensional (3D) structure of the target protein is unknown, as they rely solely on the chemical information of known active ligands [20]. In contrast, target-based methods, such as molecular docking, depend on the availability of the target's 3D structure to predict how a small molecule might bind within a specific protein binding pocket [13] [68]. With the advent of high-quality predicted protein structures from tools like AlphaFold and OmegaFold, the applicability of structure-based methods has expanded significantly [13] [68].
This guide provides a systematic performance comparison of popular tools from both categories, presenting quantitative benchmarking data, detailed experimental protocols, and practical resources to inform researchers' choices.
Performance Benchmarking of Popular Tools
Systematic Comparison of Seven Target Prediction Methods
A precise comparative study evaluated seven stand-alone codes and web servers for target prediction using a shared benchmark dataset of FDA-approved drugs to ensure a fair assessment [6]. The table below summarizes the key characteristics and findings of this benchmark.
Table 1: Systematic Comparison of Seven Target Prediction Methods [6]
| Method | Type | Core Algorithm | Key Features | Performance Notes |
|---|---|---|---|---|
| MolTarPred | Ligand-centric | 2D Similarity | Uses MACCS or Morgan fingerprints; Tanimoto or Dice scores [6]. | Most effective method in the benchmark; Morgan fingerprints with Tanimoto score recommended [6]. |
| PPB2 | Ligand-centric | Nearest Neighbor/Naïve Bayes/Deep Neural Network | Uses MQN, Xfp, and ECFP4 fingerprints; considers top 2000 similar ligands [6]. | Performance evaluated in comparative study [6]. |
| RF-QSAR | Target-centric | Random Forest | Model built from ChEMBL data using ECFP4 fingerprints [6]. | Performance evaluated in comparative study [6]. |
| TargetNet | Target-centric | Naïve Bayes | Utilizes multiple fingerprint types (FP2, MACCS, E-state, ECFP) [6]. | Performance evaluated in comparative study [6]. |
| ChEMBL | Target-centric | Random Forest | Built on ChEMBL database using Morgan fingerprints [6]. | Performance evaluated in comparative study [6]. |
| CMTNN | Target-centric | Multitask Neural Network | Uses Morgan fingerprints; run as a stand-alone code [6]. | Performance evaluated in comparative study [6]. |
| SuperPred | Ligand-centric | 2D/Fragment/3D Similarity | Based on ECFP4 fingerprints [6]. | Performance evaluated in comparative study [6]. |
The benchmark concluded that MolTarPred was the most effective method overall [6]. The study also provided crucial insights for practical application:
- Model Optimization: Using a high-confidence filter (confidence score ≥7) on interaction data improved data quality but reduced recall, making this strategy less ideal for drug repurposing where sensitivity is key [6].
- Fingerprint and Metric Selection: For MolTarPred, using Morgan fingerprints with a Tanimoto similarity score provided superior performance compared to MACCS fingerprints with a Dice score [6].
Performance of Advanced Machine Learning and Deep Learning Models
Beyond the classical tools, newer deep learning models for drug-target binding affinity (DTA) prediction have also been subject to benchmarking. The following table compiles the performance of several advanced models on established datasets like Davis and KIBA, which are standard for regression-based DTA prediction.
Table 2: Performance of Advanced DTA Prediction Models on Benchmark Datasets
| Model | Core Approach | Davis (CI) | KIBA (CI) | Key Innovation |
|---|---|---|---|---|
| DTA-GTOmega | Graph Transformer + OmegaFold Structures | 0.903 | 0.891 | Uses OmegaFold-predicted 3D protein structures and a co-attention mechanism [68]. |
| WPGraphDTA | Graph Neural Network + Word2Vec | 0.885 | 0.872 | Extracts protein features using Word2Vec on "biological words" (3-gram amino acids) [69]. |
| DeepDTA | Convolutional Neural Network (CNN) | 0.878 | 0.863 | A foundational model that processes SMILES strings and protein sequences with CNNs [69]. |
| GraphDTA | Graph Neural Network (GNN) | 0.882 | 0.868 | Represents drugs as molecular graphs instead of SMILES strings [69]. |
| KronRLS | Kernel-Based Regularized Least Squares | 0.871 | 0.857 | A classical machine learning method using drug and target similarity matrices [69]. |
| SimBoost | Gradient Boosting Machine | 0.872 | 0.860 | Uses feature engineering to create similarity-based and network-based features [69]. |
These results demonstrate that models leveraging modern architectures like graph neural networks and transformers generally outperform classical machine learning methods. Furthermore, the integration of high-quality 3D structural information, as seen in DTA-GTOmega, appears to provide a tangible performance advantage [68].
Experimental Protocols for Key Benchmarking Studies
Protocol for Benchmarking Target Prediction Methods
The methodology from the comparative study of the seven tools provides a robust template for fair performance evaluation [6].
1. Database Selection and Preparation:
- Source: The ChEMBL database (version 34) was used due to its extensive, experimentally validated bioactivity data [6].
- Data Retrieval: Bioactivity records (IC50, Ki, EC50 ≤ 10000 nM) were retrieved from relevant tables (
molecule_dictionary,target_dictionary,activities). - Data Curation:
- Non-specific or multi-protein targets were filtered out.
- Duplicate compound-target pairs were removed, resulting in 1,150,487 unique interactions.
- A high-confidence subset was created by filtering for interactions with a confidence score ≥ 7 [6].
2. Benchmark Dataset Construction:
- A set of 100 FDA-approved drugs was randomly selected from ChEMBL.
- Critical Step: These benchmark molecules were rigorously excluded from the main database used for predictions to prevent data leakage and overestimation of performance [6].
3. Target Prediction and Validation:
- Each of the seven methods was used to predict targets for the 100 benchmark drugs.
- Stand-alone codes (MolTarPred, CMTNN) were run locally, while web servers (PPB2, RF-QSAR, etc.) required manual query submission [6].
- Predictions were validated against the known interactions in the curated ChEMBL dataset, with metrics like recall being a key measure of performance, especially for drug repurposing applications [6].
Protocol for Evaluating DTA Models
Benchmarking regression-based DTA models, such as those in Table 2, follows a different protocol centered on standardized datasets and data splits.
1. Standardized Datasets:
- Davis: Contains binding affinities (Kd) for 72 kinase inhibitors against 442 kinases [68] [69].
- KIBA: Provides a unified "KIBA score" derived from Ki, Kd, and IC50 values for kinase inhibitors, designed to handle inconsistencies between different measurement types [68] [69].
- BindingDB_Kd: A subset of the BindingDB database containing dissociation constant (Kd) values [68].
2. Data Splitting Strategies: A rigorous evaluation must go beyond random splits to assess model generalizability in realistic scenarios [70].
- Random Split: Compounds and targets are randomly assigned to training and test sets. This is the least challenging scenario.
- Cold-Drug Split: All compounds in the test set are unseen during training. Tests the model's ability to generalize to novel chemotypes.
- Cold-Target Split: All targets in the test set are unseen during training. Tests the model's ability to predict for novel proteins.
- Cold-Drug-Target Split: No drug-target pair in the test set is present in the training set. This is the most challenging and realistic scenario for novel prediction [68].
3. Evaluation Metrics:
- Concordance Index (CI): Measures the ranking quality of predictions. A CI of 0.5 is random, and 1.0 is perfect ranking.
- Mean Squared Error (MSE): Measures the average squared difference between predicted and actual values.
- Regression Coefficient (r²): Represents the proportion of variance in the affinity values explained by the model.
Workflow Visualization of Benchmarking and Prediction Strategies
The following diagram illustrates the logical workflow for selecting a DTI prediction strategy based on data availability, a central concept in chemogenomics.
Figure 1: Decision workflow for selecting a DTI prediction strategy.
The benchmarking data reveals that an integrated approach, leveraging both ligand and target information, often yields the most robust results. The following workflow is adapted from real-world studies that combine multiple methods to improve hit identification and prioritization [6] [20].
Figure 2: An integrated virtual screening workflow combining ligand- and structure-based methods.
Successful implementation and benchmarking of DTI prediction tools rely on a foundation of key public databases and software resources.
Table 3: Essential Resources for DTI Prediction Research
| Resource Name | Type | Primary Function | Relevance to Benchmarking |
|---|---|---|---|
| ChEMBL | Database | Manually curated database of bioactive molecules with drug-like properties [6] [70]. | Primary source of reliable bioactivity data for training and benchmarking target-centric and ligand-centric models [6]. |
| BindingDB | Database | Public database of measured binding affinities for drug-target interactions [68]. | Provides binding affinity data (Kd, Ki) essential for training and evaluating regression-based DTA models [68]. |
| RDKit | Software | Open-source cheminformatics toolkit [68] [71]. | Used for processing SMILES, generating molecular fingerprints and descriptors, and calculating molecular properties [68]. |
| AlphaFold/OmegaFold | Software | Protein structure prediction tools [13] [68]. | Provides high-quality 3D protein structures for target-based methods when experimental structures are unavailable, expanding target coverage [68]. |
| MolScore | Software | Scoring and benchmarking framework for generative models and de novo molecular design [71]. | Provides a suite of drug-relevant scoring functions (e.g., docking, QSAR) and performance metrics to standardize model evaluation [71]. |
The accurate prediction of Drug-Target Interactions (DTIs) represents a critical challenge in modern drug discovery, with implications for target identification, drug repurposing, and polypharmacology studies [6] [13]. Chemogenomic approaches, which simultaneously utilize both chemical and genomic information, have emerged as powerful computational frameworks for addressing this challenge [72]. These methods can be broadly categorized into two complementary paradigms: ligand-based and target-based approaches. Ligand-based methods operate on the principle that similar compounds tend to interact with similar protein targets, relying primarily on chemical similarity metrics and known ligand information [6] [73]. In contrast, target-based approaches leverage structural or sequence information about the protein targets, often employing molecular docking or structure-based similarity measures [6] [74]. As both strategies continue to evolve, a systematic comparison of their performance metrics—including accuracy, enrichment capability, and reliability—becomes essential for guiding methodological selection and advancement in computational drug discovery. This review provides a comprehensive performance comparison of these approaches, supported by experimental data and standardized evaluation protocols.
Performance Metrics and Benchmarking Frameworks
The evaluation of chemogenomic methods requires multiple performance metrics to provide a comprehensive view of model capabilities. Accuracy measures the overall correctness of predictions, while enrichment assesses the ability to prioritize true interactions early in the ranking process, which is particularly crucial for virtual screening applications [75]. Reliability refers to the consistency and confidence of predictions across different targets and chemical spaces [73].
Standardized benchmarking datasets and protocols have been established to enable fair comparisons. The Yamanishi dataset serves as a "golden standard" in DTI research, providing curated interactions across different protein families like GPCRs, kinases, nuclear receptors, and ion channels [72] [76]. For structure-based approaches, benchmark sets like CASF-2016, DUD-E, and LIT-PCBA provide decoy molecules and standardized protocols to evaluate enrichment performance [75]. The ProSPECCTs collection offers datasets specifically designed to evaluate pocket comparison approaches under various scenarios, enabling systematic assessment of binding site similarity methods [74].
Table 1: Key Performance Metrics for DTI Prediction Evaluation
| Metric Category | Specific Metrics | Definition and Significance |
|---|---|---|
| Overall Accuracy | AUC-ROC | Area Under the Receiver Operating Characteristic curve; measures overall classification performance |
| AUC-PR | Area Under the Precision-Recall curve; more informative for imbalanced datasets | |
| Accuracy | Overall proportion of correct predictions | |
| Enrichment Power | Top 1% EF | Enrichment Factor in the top 1% of ranked molecules; measures early recognition capability |
| Recall | Proportion of true positives identified from all actual positives | |
| Precision | Proportion of true positives among all predicted positives | |
| Reliability | Confidence Scores | Quantitative measures of prediction reliability [73] |
| Similarity Thresholds | Fingerprint-dependent thresholds for filtering background noise [73] |
Performance Comparison of Ligand-Based and Target-Based Methods
Accuracy Metrics
A systematic comparison of seven target prediction methods using a shared benchmark dataset of FDA-approved drugs revealed significant performance differences between approaches [6]. The study evaluated both stand-alone codes and web servers, including MolTarPred, PPB2, RF-QSAR, TargetNet, ChEMBL, CMTNN, and SuperPred. Among these, the ligand-based method MolTarPred demonstrated superior performance, particularly when using Morgan fingerprints with Tanimoto scores, which outperformed MACCS fingerprints with Dice scores [6].
The accuracy of ligand-based methods is highly dependent on the chemical similarity metrics and fingerprint representations employed. Research has shown that the distribution of effective similarity scores for target fishing is fingerprint-dependent, with optimal similarity thresholds varying significantly across different fingerprint types [73]. For instance, ECFP4 and FCFP4 fingerprints generally provide better performance for target prediction compared to simpler fingerprints like MACCS [73].
Target-based approaches, particularly those utilizing molecular docking, face challenges with scoring function accuracy. Traditional docking scoring functions typically achieve Pearson correlation coefficients of only 0.2 to 0.5 between predicted binding affinities and experimental values [75]. However, recent machine learning-based scoring functions have demonstrated significant improvements, with some models achieving correlation coefficients exceeding 0.8 [75].
Enrichment Factors
Enrichment capability is particularly important for practical virtual screening applications where the goal is to identify active compounds from large chemical libraries. The performance gap between accuracy-oriented models and enrichment-oriented models can be substantial.
Advanced hybrid models that combine graph neural networks with physics-based scoring methods have demonstrated remarkable enrichment capabilities. The AK-Score2 model achieved top 1% enrichment factors of 32.7 and 23.1 with the CASF-2016 and DUD-E benchmark sets, respectively, outperforming most existing methods in forward screening [75]. This performance highlights the potential of integrating multiple complementary approaches.
Ligand-based methods also show strong enrichment potential, particularly when applying appropriate similarity thresholds. Studies have demonstrated that using fingerprint-dependent similarity thresholds can significantly enhance the confidence of enriched targets by filtering out background noise, thereby improving both precision and recall [73].
Reliability and Confidence Assessment
The reliability of predictions varies considerably between approaches and is influenced by multiple factors. For ligand-based methods, the similarity between the query molecule and reference ligands serves as a crucial indicator of confidence [73]. Setting appropriate similarity thresholds that are fingerprint-specific can significantly enhance reliability by balancing precision and recall.
Target-based methods face reliability challenges related to protein structural data quality and coverage. The emergence of AlphaFold-predicted structures has dramatically expanded the structural coverage of human proteomes, with over 32,000 druggable pockets identified across 20,000 protein domains using experimentally determined structures and AlphaFold2 models [74]. However, the reliability of predictions using computational structures may vary compared to those using experimental structures.
Table 2: Comparative Performance of Representative Methods
| Method | Approach Type | Key Features | Reported Performance |
|---|---|---|---|
| MolTarPred [6] | Ligand-based | 2D similarity using Morgan fingerprints | Most effective in systematic comparison |
| AK-Score2 [75] | Target-based Hybrid | Graph neural networks + physics-based scoring | Top 1% EF: 32.7 (CASF-2016), 23.1 (DUD-E) |
| PocketVec [74] | Target-based | Inverse virtual screening of lead-like molecules | Comparable to leading methodologies with wider applicability |
| SVDTI [72] | Hybrid | Stacked variational autoencoder with SMILES and protein sequences | Remarkable improvements vs. state-of-the-art methods |
| RNIDTP [76] | Ligand-based | Reliable negative sample selection + feature selection | Superior to random negative sample selection |
Experimental Protocols and Methodologies
Benchmarking Experimental Setup
Standardized benchmarking is essential for fair performance comparison. A robust experimental protocol should include:
Dataset Preparation: The ChEMBL database (version 34) provides comprehensive bioactivity data, containing 15,598 targets, 2,431,025 compounds, and 20,772,701 interactions [6]. To ensure data quality, filtering criteria should include:
- Selection of bioactivity records with standard values (IC50, Ki, or EC50) below 10,000 nM
- Exclusion of entries associated with non-specific or multi-protein targets
- Removal of duplicate compound-target pairs
- Application of confidence scores (e.g., minimum score of 7 in ChEMBL) to retain only well-validated interactions [6]
Performance Evaluation: Using a shared benchmark dataset of FDA-approved drugs with molecules excluded from the main database to prevent overlap and biased performance estimation [6]. Typically, 100 random samples are sufficient for validation [6].
Validation Metrics: Implementation of rigorous validation metrics including AUC-ROC, AUC-PR, enrichment factors, and precision-recall curves under different similarity thresholds [73].
Ligand-Based Method Workflow
Ligand-Based Target Fishing Workflow
The ligand-based target fishing workflow involves several key stages:
Reference Library Construction: Curate a high-quality library from sources like ChEMBL and BindingDB, containing protein targets and their associated ligands with strong bioactivity (IC50, Ki, Kd, or EC50 < 1 μM) [73]. The library should include diverse target categories: enzymes, membrane receptors, ion channels, and transporters.
Fingerprint Calculation: Compute multiple two-dimensional fingerprint representations for each compound using tools like RDKit [73]. Key fingerprint types include:
- ECFP4/FCFP4: Circular fingerprints capturing atom environments
- AtomPair: Based on topological atom pairs
- MACCS: Structural key fingerprints using predefined substructures
- Avalon, RDKit, Layered, and Torsion fingerprints
Similarity Calculation and Threshold Application: For a query compound, perform pairwise similarity searching against the reference library using the Tanimoto coefficient [73]. Apply fingerprint-specific similarity thresholds to filter background noise and enhance confidence.
Target Ranking and Confidence Assessment: Rank potential targets based on similarity scores and apply ensemble methods where multiple fingerprints are integrated to improve reliability [73].
Target-Based Method Workflow
Target-Based Binding Site Analysis
Structure-based approaches follow this general protocol:
Pocket Identification: Detect druggable pockets in protein structures using algorithms like PocketVec, which can work with both experimental structures (from PDB) and predicted structures (from AlphaFold2) [74].
Probe Molecule Selection: Curate a diverse set of small molecules for inverse screening. Two common approaches include:
Inverse Virtual Screening: Use molecular docking programs (rDock for rigid docking or SMINA for flexible docking) to assess potential binding of probe molecules to identified pockets [74].
Descriptor Generation and Comparison: Convert docking scores into rankings stored in vector-type descriptors (PocketVec), enabling similarity comparisons between binding sites [74].
Table 3: Key Research Resources for DTI Prediction Studies
| Resource Category | Specific Tools/Databases | Function and Application |
|---|---|---|
| Bioactivity Databases | ChEMBL [6] [73] | Manually curated database of bioactive molecules with drug-like properties |
| BindingDB [73] | Database of measured binding affinities focusing on drug-target interactions | |
| PubChem BioAssay [73] | Repository of biological activity data from high-throughput screening | |
| Compound Libraries | Glide Chemically Diverse Collection [74] | Fragment-like molecules (50-200 g·mol⁻¹) for inverse virtual screening |
| MOE Lead-like Molecules [74] | Lead-like compounds (200-450 g·mol⁻¹) for structure-based screening | |
| Software Tools | RDKit [73] | Open-source cheminformatics toolkit for fingerprint calculation and similarity searching |
| AutoDock-GPU [75] | Molecular docking software for generating decoy structures and binding poses | |
| rDock & SMINA [74] | Docking programs for rigid and flexible molecular docking, respectively | |
| Benchmark Datasets | Yamanishi Dataset [72] [76] | Golden standard dataset for DTI prediction across protein families |
| CASF-2016, DUD-E, LIT-PCBA [75] | Benchmark sets for evaluating enrichment factors in virtual screening | |
| ProSPECCTs [74] | Dataset collection for evaluating pocket comparison approaches |
The comprehensive comparison of ligand-based and target-based chemogenomic approaches reveals a complex performance landscape where each paradigm offers distinct advantages. Ligand-based methods excel when similar reference ligands are available in comprehensive databases, with performance highly dependent on fingerprint selection and similarity thresholds [6] [73]. Target-based approaches provide value when structural information is available and reliable, particularly for novel targets with limited known ligands [6] [74].
The emerging trend toward hybrid models that integrate both ligand and target information shows promising performance improvements [75] [72]. Methods like AK-Score2 that combine graph neural networks with physics-based scoring demonstrate exceptional enrichment factors, while approaches like SVDTI that integrate stacked variational autoencoders with collaborative filtering show enhanced predictive accuracy [75] [72].
Future methodological development should focus on several key areas: (1) improving the handling of data sparsity through better negative sample selection algorithms [76], (2) enhancing reliability assessment with quantitative confidence scores [73], (3) expanding proteome coverage through integration of AlphaFold-predicted structures [74], and (4) developing standardized benchmarking protocols that enable fair comparison across diverse methodological frameworks [6] [75].
As the field advances, the integration of multiple complementary approaches—leveraging the strengths of both ligand-based and target-based strategies—appears most promising for achieving robust, accurate, and reliable drug-target interaction predictions that can effectively accelerate drug discovery pipelines.
The systematic identification of drug-target interactions (DTIs) is a fundamental pillar of modern drug discovery. In recent decades, the paradigm has shifted from traditional receptor-specific studies to a comprehensive cross-receptor view, giving rise to the interdisciplinary field of chemogenomics [3]. This field attempts to derive predictive links between the chemical structures of bioactive molecules and the receptors with which they interact, accelerating the discovery of novel chemical starting points for drug development programs. Chemogenomic approaches broadly fall into two principal categories: ligand-based and target-based methods, each with distinct philosophical underpinnings and methodological frameworks [6] [43].
Ligand-based methods operate on the principle that similar molecules tend to exhibit similar biological activities and bind to similar protein targets [11]. These approaches rely heavily on the knowledge of known ligands and their annotated targets, using molecular similarity calculations to predict new interactions. In contrast, target-based methods, often referred to as structure-based methods, build predictive models for each target. These frequently use the three-dimensional (3D) structure of the target protein to estimate whether a query molecule is likely to interact via techniques like molecular docking [6] [77]. A third, emerging category is the hybrid approach, which integrates both ligand and target information into a single model, often using proteochemometric (PCM) modeling or machine learning techniques that simultaneously process features of both compounds and proteins [78] [79].
This guide provides an objective comparison of these approaches, detailing their respective strengths, weaknesses, and ideal applications to aid researchers in selecting the most appropriate strategy for their drug discovery projects.
Comparative Analysis of Approaches
The following table provides a systematic comparison of the core chemogenomic approaches, synthesizing information from recent evaluations and methodological reviews.
Table 1: Comparative analysis of ligand-based, target-based, and hybrid chemogenomic approaches.
| Feature | Ligand-Based Approaches | Target-Based Approaches | Hybrid/Chemogenomic Approaches |
|---|---|---|---|
| Core Principle | "Similar ligands bind similar targets" [3] [11] | Predictive models built for each target, often based on 3D structure [6] | Integrates both ligand and target descriptors into a single model [78] [79] |
| Data Requirements | Known active ligands (chemical structures, bioactivity) [6] | Target structure (X-ray, NMR, or AlphaFold model) or bioactivity data for QSAR [6] [77] | Chemical structures of ligands and amino acid sequences/structures of targets [11] [79] |
| Key Strengths | - High speed, suitable for large library screening [77] [78]- No need for target 3D structure [43]- High interpretability through chemical similarity [43] | - Can find novel chemotypes unrelated to known ligands [77]- Provides a structural model of binding (pose) [77] | - Can predict interactions for new targets & new compounds [79]- Identifies off-target effects & enables polypharmacology studies [6] [79] |
| Key Weaknesses | - Cannot find novel scaffolds (scaffold hopping is difficult) [77]- Fails if few ligands are known ("cold start" problem) [43] | - Limited by availability of high-quality 3D structures [6] [77]- Computationally intensive [77]- Handling protein flexibility is challenging [77] [78] | - Risk of over-optimistic performance with random data splitting [79]- Models may over-rely on compound features due to data bias [79] |
| Ideal Use Cases | - Target fishing for compounds with known analoges [6]- Early-stage virtual screening when target structure is unknown [78] | - Structure-based lead optimization- Targets with no known ligands but a known structure [77] | - Proteochemometric (PCM) modeling for diverse target families [79]- Large-scale drug repurposing and side-effect prediction [79] |
Experimental Protocols and Benchmarking
Robust experimental design and benchmarking are critical for the objective evaluation of different chemogenomic methods. Recent studies have established rigorous protocols to ensure fair and realistic performance assessments.
Database Preparation and Curation
A critical first step involves the compilation of high-quality, non-redundant interaction data. A typical protocol, as used in a 2025 benchmark study, involves:
- Source Selection: Utilizing experimentally validated databases like ChEMBL (version 34 was used, containing over 2.4 million compounds and 2 million interactions) or DrugBank [6] [11].
- Data Retrieval: Querying relevant tables (e.g.,
molecule_dictionary,target_dictionary,activities) to retrieve canonical SMILES strings for compounds, target identifiers, and bioactivity values (e.g., IC50, Ki, EC50) [6]. - Filtering: Applying a confidence score filter (e.g., a minimum of 7 in ChEMBL, indicating a direct assigned target) to ensure only high-confidence interactions are included. Entries associated with non-specific or multi-protein targets are excluded [6].
- Deduplication: Removing duplicate compound-target pairs to retain only unique interactions for analysis [6].
Benchmark Dataset Construction
To evaluate target prediction methods fairly, a benchmark dataset of FDA-approved drugs is often prepared. The key is to ensure that these molecules are excluded from the main database used for prediction to prevent overestimation of performance. Typically, 100 or more random samples of FDA-approved drugs are selected as query molecules, and the remaining molecules form the database for identifying potential interactions [6].
Critical Consideration: Data Splitting Strategy
A pivotal finding in recent machine learning-based DTI prediction is that the method of splitting data into training and test sets drastically impacts performance evaluation [79].
- Random Splitting: Leads to near-complete data memorization and produces highly over-optimistic results because structurally similar compounds related to the test set are present in the training data. This practice should be avoided for realistic model assessment [79].
- Network-Based or Cold-Start Splitting: These are recommended strategies. They involve splitting the data so that the test set contains drugs or targets (or both) that are not seen during training, which provides a more challenging and realistic estimate of a model's predictive power in a real-world scenario [79].
Performance Evaluation
After training models on the training set, their performance is evaluated on the held-out test set. Common metrics include:
- Recall (Sensitivity): The proportion of actual positives that are correctly identified. This is crucial for drug repurposing, where missing a potential interaction (false negative) is undesirable [6].
- Precision: The proportion of positive identifications that are actually correct. High precision is important to reduce the cost of experimental validation of false positives.
- Enrichment Factor: Measures the concentration of true active compounds in a screened subset compared to a random selection.
The following diagram illustrates the workflow for a rigorous comparative benchmark study.
Figure 1: Workflow for benchmarking drug-target interaction prediction methods.
Successful implementation of chemogenomic approaches relies on a suite of publicly available databases and software tools. The table below details essential resources for building and validating predictive models.
Table 2: Key research reagents and resources for chemogenomic research.
| Resource Name | Type | Primary Function | Relevance |
|---|---|---|---|
| ChEMBL [6] [11] | Database | A manually curated database of bioactive molecules with drug-like properties, containing binding affinities, functional assays, and ADMET data. | Primary source of high-quality, experimentally validated bioactivity data for training and benchmarking ligand-based and hybrid models. |
| DrugBank [11] [79] | Database | A comprehensive database containing detailed drug and drug target information, including FDA-approved drug products. | Essential for drug repurposing studies and for constructing benchmark datasets of approved drugs. |
| AlphaFold [6] | Software/Database | Provides highly accurate protein structure predictions for proteins with unknown experimental 3D structures. | Expands the scope of target-based methods by providing reliable structural models for targets that lack crystal structures. |
| MolTarPred [6] | Software/Web Server | A ligand-based target prediction method that uses 2D similarity searching against known ligands in ChEMBL. | Identified as one of the most effective methods in a recent benchmark; useful for generating MoA hypotheses and drug repurposing. |
| AutoDock Vina [77] | Software | A widely used program for molecular docking, predicting how small molecules bind to a macromolecular target. | A standard tool for structure-based virtual screening in target-based approaches. |
| Proteochemometric (PCM) Modeling [79] | Methodology | A machine learning framework that uses both compound and target protein descriptors to predict interactions under a single model. | The foundational methodology for modern hybrid chemogenomic approaches, enabling prediction for new compounds and new targets. |
The comparative analysis presented in this guide reveals that the choice between ligand-based, target-based, and hybrid chemogenomic approaches is not a matter of identifying a single superior method, but rather of selecting the right tool for the specific research question and available data. Ligand-based methods offer speed and simplicity when known active ligands exist, while target-based approaches can unlock novel chemotypes when a reliable protein structure is available. The emerging hybrid and PCM models, powered by machine learning, offer a powerful integrative framework that is particularly well-suited for large-scale drug repurposing and the systematic exploration of polypharmacology.
Future advancements in this field will likely be driven by more sophisticated protein featurization techniques, such as protein language models [79], and a stronger emphasis on rigorous benchmarking using network-aware data splitting strategies to ensure predictive models deliver real-world value. By understanding the strengths and limitations of each paradigm, researchers can more effectively navigate the complex landscape of drug-target interaction prediction.
The paradigm of drug discovery has progressively shifted from a traditional "one drug, one target" approach toward a more holistic systems pharmacology strategy that embraces polypharmacology and multi-target drug discovery [80]. This transformation is largely defined by the flood of data on ligand properties and binding to therapeutic targets, abundant computing capacities, and the advent of on-demand virtual libraries of drug-like small molecules in their billions [81]. Within this landscape, chemogenomic approaches have emerged as powerful computational methods that integrate chemical and biological information to predict drug-target interactions (DTIs).
These approaches primarily fall into two categories: ligand-based methods, which leverage similarity between known and query compounds, and target-based methods, which utilize protein structures or sequences to model interactions. However, the predictive power of these in silico models remains contingent upon rigorous experimental validation in vitro to confirm biological relevance and therapeutic potential. This review systematically compares ligand-based and target-based chemogenomic approaches, examines their performance characteristics, and underscores the indispensable role of experimental validation in translating computational predictions into biologically meaningful outcomes.
Comparative Analysis of Chemogenomic Approaches
Fundamental Principles and Methodologies
Ligand-based approaches operate on the similar property principle, which posits that structurally similar molecules are likely to exhibit similar biological activities [6] [11]. These methods rely on chemical similarity searching against comprehensive databases of known bioactive compounds, such as ChEMBL and DrugBank. The effectiveness of ligand-based methods hinges on the knowledge of known ligands and their annotated targets [6]. Key techniques include similarity searching using molecular fingerprints (e.g., ECFP, Morgan fingerprints) and machine learning models trained on chemical structures [6].
In contrast, target-based approaches leverage structural or sequence information about the protein target. Structure-based methods utilize molecular docking to predict how small molecules interact with protein binding sites, while target-centric machine learning models build predictive models for each target using various algorithms [6] [82]. These approaches have been significantly advanced by computational tools like AlphaFold, which can generate high-quality structural models from amino acid sequences even without experimental determination [6].
Performance Comparison of Representative Methods
A precise comparison of seven target prediction methods published in 2025 revealed significant performance variations between ligand-based and target-based approaches [6]. The study evaluated stand-alone codes and web servers using a shared benchmark dataset of FDA-approved drugs, with results summarized in the table below.
Table 1: Performance Comparison of Representative Target Prediction Methods
| Method | Type | Source | Algorithm | Key Features |
|---|---|---|---|---|
| MolTarPred | Ligand-based | ChEMBL 20 | 2D similarity | MACCS fingerprints; Top 1,5,10,15 similar ligands [6] |
| PPB2 | Ligand-based | ChEMBL 22 | Nearest neighbor/Naïve Bayes/deep neural network | MQN, Xfp and ECFP4 fingerprints; Top 2000 similar ligands [6] |
| SuperPred | Ligand-based | ChEMBL and BindingDB | 2D/fragment/3D similarity | ECFP4 fingerprints [6] |
| RF-QSAR | Target-centric | ChEMBL 20&21 | Random forest | ECFP4 fingerprints; Top 4,7,11,33,66,88,110 [6] |
| TargetNet | Target-centric | BindingDB | Naïve Bayes | FP2, Daylight-like, MACCS, E-state and ECFP2/4/6 fingerprints [6] |
| ChEMBL | Target-centric | ChEMBL 24 | Random forest | Morgan fingerprints [6] |
| CMTNN | Target-centric | ChEMBL 34 | ONNX runtime | Morgan fingerprints [6] |
| DeepDTAGen | Multitask DL | KIBA, Davis, BindingDB | Multitask deep learning | Predicts binding affinity & generates novel drugs [83] |
The analysis demonstrated that MolTarPred emerged as the most effective method among those evaluated [6]. The study also explored model optimization strategies, revealing that for MolTarPred, Morgan fingerprints with Tanimoto scores outperformed MACCS fingerprints with Dice scores [6]. However, the implementation of high-confidence filtering, while improving precision, reduced recall, making it less ideal for drug repurposing applications where maximizing potential hit identification is crucial [6].
Table 2: Quantitative Performance Metrics Across Method Types
| Method Category | Representative Performance | Strengths | Limitations |
|---|---|---|---|
| Ligand-based | MolTarPred: Highest effectiveness in benchmark [6] | Effective when known ligands exist; Fast screening; No protein structure needed [6] | Limited to targets with known ligands; Cannot discover novel scaffolds [11] |
| Target-based | DeepDTAGen: MSE 0.146, CI 0.897 on KIBA [83] | Can identify novel chemotypes; Structure-guided design [82] | Dependent on quality of protein structures; Higher computational cost [6] |
| Hybrid Models | Emerging multitask frameworks [83] | Leverages both chemical and structural information; More comprehensive predictions [83] | Implementation complexity; Data integration challenges [83] |
Advanced multitask learning frameworks like DeepDTAGen represent the next evolution in chemogenomic approaches, simultaneously predicting drug-target binding affinities and generating novel target-aware drug variants using common features for both tasks [83]. On the KIBA dataset, DeepDTAGen achieved a mean squared error (MSE) of 0.146, concordance index (CI) of 0.897, and ({r}_{m}^{2}) of 0.765, outperforming traditional single-task models [83].
Experimental Validation: From Computational Prediction to Biological Confidence
Essential Validation Methodologies
The transition from in silico predictions to biologically relevant outcomes requires robust experimental validation methodologies that confirm computational findings in physiologically relevant systems.
Cellular Thermal Shift Assay (CETSA) has emerged as a leading approach for validating direct target engagement in intact cells and native tissue environments [84]. This method detects changes in protein thermal stability induced by ligand binding, providing direct evidence of compound-target interactions within complex biological systems. Recent work by Mazur et al. (2024) applied CETSA in combination with high-resolution mass spectrometry to quantify drug-target engagement of DPP9 in rat tissue, confirming dose- and temperature-dependent stabilization ex vivo and in vivo [84].
In vitro binding assays remain fundamental for validating predicted interactions. These include:
- Binding affinity assays to determine dissociation constants (Kd)
- Enzyme inhibition assays to measure inhibitory concentrations (IC50)
- Cell-based viability assays to assess functional biological effects
For example, in the discovery of PKMYT1 inhibitors for pancreatic cancer, researchers conducted in vivo experiments showing that the computationally identified compound HIT101481851 inhibited the viability of pancreatic cancer cell lines in a dose-dependent manner while exhibiting lower toxicity toward normal pancreatic epithelial cells [82].
Structural validation techniques including X-ray crystallography and cryo-electron microscopy provide atomic-level insights into protein-ligand interactions, enabling rational optimization of binding characteristics [81]. These methods are particularly valuable for confirming binding modes predicted by molecular docking studies.
Integrated Workflow for Validation
The following diagram illustrates a comprehensive workflow integrating computational prediction with experimental validation:
Diagram 1: Integrated Drug Discovery Workflow
This integrated workflow emphasizes the iterative nature of modern drug discovery, where computational predictions inform experimental design, and experimental results feedback to refine computational models.
Case Studies: Successful Integration of In Silico and In Vitro Approaches
PKMYT1 Inhibitor Discovery for Pancreatic Cancer
A recent study exemplifies the successful application of structure-based discovery followed by experimental validation [82]. Researchers implemented a comprehensive pipeline to identify novel PKMYT1 inhibitors, a validated therapeutic target in pancreatic cancer. The computational phase included:
- Pharmacophore modeling using four PKMYT1 co-crystal structures (PDB ID 8ZTX, 8ZU2, 8ZUD, and 8ZUL)
- Virtual screening of 1.64 million compounds from the TargetMol natural compound library
- Molecular docking using Glide module with HTVS, SP, and XP precision modes
- Molecular dynamics simulations of 1 μs to confirm binding stability
This integrated computational approach identified HIT101481851 as a promising candidate with favorable binding characteristics and stable interactions with key residues such as CYS-190 and PHE-240 [82]. Experimental validation confirmed that HIT101481851 inhibited pancreatic cancer cell viability in a dose-dependent manner while exhibiting lower toxicity toward normal pancreatic epithelial cells [82].
Drug Repurposing through Ligand-Based Prediction
The ligand-based method MolTarPred demonstrated the potential for drug repurposing through successful prediction of novel drug-target interactions [6]. A case study on fenofibric acid showed its potential for drug repurposing as a THRB modulator for thyroid cancer treatment [6]. In another example, MolTarPred discovered hMAPK14 as a potent target of mebendazole, which was further validated through in vitro experiments [6]. Similarly, MolTarPred predicted Carbonic Anhydrase II (CAII) as a new target of Actarit, suggesting potential for repurposing this rheumatoid arthritis drug for conditions such as hypertension, epilepsy, and certain cancers [6].
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Research Reagents and Platforms for Experimental Validation
| Reagent/Platform | Function | Application in Validation |
|---|---|---|
| CETSA | Cellular target engagement validation | Confirms direct binding in physiologically relevant cellular environments [84] |
| Schrödinger Suite | Integrated drug discovery platform | Protein preparation, molecular docking, dynamics simulations [82] |
| Desmond MD System | Molecular dynamics simulation | Analyzes protein-ligand complex stability over time [82] |
| OPLS4 Force Field | Molecular mechanics parameterization | Energy minimization and structural validation [82] |
| TargetMol Compound Library | Collection of bioactive molecules | Source of diverse chemical matter for virtual screening [82] |
| MO:BOT Platform | Automated 3D cell culture | Standardizes organoid production for biologically relevant screening [85] |
| eProtein Discovery System | Automated protein production | Accelerates from DNA to purified protein for functional studies [85] |
| Firefly+ Platform | Automated liquid handling | Standardizes genomic workflows and enhances reproducibility [85] |
The integration of ligand-based and target-based chemogenomic approaches represents a powerful strategy for modern drug discovery. While each approach has distinct strengths and limitations, their complementary nature enables more comprehensive exploration of chemical and target space. Ligand-based methods excel in leveraging existing chemical knowledge for applications like drug repurposing, while target-based approaches enable novel chemotype identification and structure-guided optimization.
Critically, the ultimate value of both approaches remains dependent on rigorous experimental validation using methodologies such as CETSA, cellular binding assays, and functional phenotypic screens. As multitask learning frameworks and hybrid models continue to evolve, the field moves closer to a unified discovery paradigm that seamlessly integrates computational prediction with experimental confirmation. This convergence, supported by advances in automation, AI, and human-relevant model systems, promises to accelerate the delivery of novel therapeutics for complex diseases while reducing attrition in the drug development pipeline.
The future of drug discovery lies not in choosing between computational or experimental approaches, but in strategically integrating both to build a continuous cycle of prediction, validation, and refinement that progressively enhances our understanding of the complex interplay between chemical compounds and biological systems.
Conclusion
The comparative analysis of ligand-based and target-based chemogenomic approaches reveals that they are not mutually exclusive but are highly complementary. Ligand-based methods offer speed and applicability when structural data is scarce, while target-based approaches provide atomic-level insight into binding mechanisms. The future of the field lies in the intelligent integration of these paradigms, further empowered by machine learning, large language models, and high-quality predicted protein structures from tools like AlphaFold. Moving forward, the key to accelerated drug discovery will be the development of robust, interpretable, and hybrid models that can seamlessly leverage both ligand information and target structures to navigate the complex landscape of polypharmacology and deliver safer, more effective multi-target therapeutics.
References
- 1. Chemogenomic approaches to rational drug design - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC1978269/]
- 2. - Wikipedia Chemogenomics [https://en.wikipedia.org/wiki/Chemogenomics]
- 3. Chemogenomic approaches to drug discovery: similar ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1978276/]
- 4. MOST: most-similar ligand based approach to target prediction [https://bmcbioinformatics.biomedcentral.com/articles/10.1186/s12859-017-1586-z]
- 5. Target fishing: from “needle in haystack” to “precise guidance” [https://pmc.ncbi.nlm.nih.gov/articles/PMC12635624/]
- 6. A precise comparison of molecular target prediction methods [https://pubs.rsc.org/en/content/articlehtml/2025/dd/d5dd00199d]
- 7. Ligand and structure-based virtual screening approaches ... [https://pubmed.ncbi.nlm.nih.gov/39223358/]
- 8. A chemogenomics view on protein-ligand spaces [https://bmcbioinformatics.biomedcentral.com/articles/10.1186/1471-2105-10-S6-S13]
- 9. Ligand-based approach for predicting drug targets and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7929377/]
- 10. Current computational methods for predicting protein ... [https://www.sciencedirect.com/science/article/pii/S2001037019301059]
- 11. A comparative chemogenic analysis for predicting Drug ... [https://www.nature.com/articles/s41598-020-63842-7]
- 12. 2.3. Target—ligand-based approaches [https://bio-protocol.org/exchange/minidetail?id=4399996&type=30]
- 13. In silico methods for drug-target interaction prediction - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC12570315/]
- 14. Prediction of drug target interaction based on under sampling ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0318420]
- 15. Drug–target interaction prediction by integrating ... [https://bmcbioinformatics.biomedcentral.com/articles/10.1186/s12859-024-05976-3]
- 16. Augmented BindingNet dataset for enhanced ligand ... [https://www.nature.com/articles/s44386-024-00003-0]
- 17. AI-Driven Polypharmacology in Small-Molecule Drug Discovery [https://pmc.ncbi.nlm.nih.gov/articles/PMC12295758/]
- 18. Polypharmacology: new drugs in 2023–2024 [https://link.springer.com/article/10.1007/s43440-025-00715-8]
- 19. : an emerging strategy for rapid Chemogenomics and drug... target [https://www.nature.com/articles/nrg1317?error=cookies_not_supported]
- 20. Leveraging ligand-based and structure-based approaches ... [https://www.iptonline.com/articles/leveraging-ligand-based-and-structure-based-approaches-for-advan]
- 21. ChEMBL [https://www.ebi.ac.uk/chembl/]
- 22. - DrugBank Commons Database [https://ngdc.cncb.ac.cn/databasecommons/database/id/296]
- 23. : a comprehensive resource for in silico DrugBank discovery and... drug [https://pmc.ncbi.nlm.nih.gov/articles/PMC1347430/]
- 24. RCSB PDB: Homepage [https://www.rcsb.org/]
- 25. Fifteen years of ChEMBL and its role in cheminformatics and ... [https://jcheminf.biomedcentral.com/articles/10.1186/s13321-025-00963-z]
- 26. Release Version 5.1.13 | DrugBank Online DrugBank [https://go.drugbank.com/releases/latest]
- 27. A new chapter for RCSB Protein Data Bank Molecule of the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12009144/]
- 28. Incorporating targeted protein structure in deep learning ... [https://pubs.rsc.org/en/content/articlehtml/2025/sc/d5sc05748e?page=search]
- 29. A comprehensive review of feature based methods for drug ... [https://www.sciencedirect.com/science/article/pii/S1532046419300772]
- 30. Recent advances in molecular representation methods and ... [https://www.nature.com/articles/s44386-025-00017-2]
- 31. AI-Integrated QSAR Modeling for Enhanced Drug Discovery [https://pmc.ncbi.nlm.nih.gov/articles/PMC12525248/]
- 32. Ligand-based pharmacophore modeling and QSAR ... [https://www.frontiersin.org/journals/molecular-biosciences/articles/10.3389/fmolb.2023.1106128/full]
- 33. Comparative Study of (Q)SAR Models for Predicting the ... [https://www.sciencedirect.com/science/article/pii/S3050620425000491]
- 34. A pharmacophore-guided deep learning approach for ... [https://www.nature.com/articles/s41467-023-41454-9]
- 35. One size does not fit all: revising traditional paradigms for ... [https://jcheminf.biomedcentral.com/articles/10.1186/s13321-025-00948-y]
- 36. Experimental and computational models to understand ... [https://www.sciencedirect.com/science/article/pii/S2772417424000050]
- 37. Harnessing free energy calculations for kinome-wide ... [https://www.nature.com/articles/s41467-025-62722-w]
- 38. Benchmarking the Structure-Based Virtual Screening ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12363558/]
- 39. Decoding the limits of deep learning in molecular docking for ... [https://pubs.rsc.org/en/content/articlehtml/2025/sc/d5sc05395a]
- 40. A Database for Large-Scale Docking and Experimental ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12226175/]
- 41. A quantitative model of structure-based virtual screening ... [https://chemrxiv.org/engage/chemrxiv/article-details/69037e08113cc7cfff050553]
- 42. FEP in 2025: We're Going to Need a Bigger Drug Discovery ... [https://cresset-group.com/science/science-resources/fep-in-2025/]
- 43. Chemogenomic Approaches for Revealing Drug Target ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8844939/]
- 44. Predicting drug-target interactions using machine learning ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12134243/]
- 45. Enhancing drug-target interaction prediction with graph ... [https://www.frontiersin.org/journals/bioinformatics/articles/10.3389/fbinf.2025.1649337/full]
- 46. Drug–target interaction prediction based on protein ... [https://www.nature.com/articles/s41598-023-30026-y]
- 47. Strategies, Challenges and Drug Repurposing Successes [https://www.technologynetworks.com/drug-discovery/articles/drug-repurposing-strategies-challenges-and-successes-384263]
- 48. Frontiers | Editorial: Opportunities and challenges in drug repurposing [https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2025.1709217/full]
- 49. AΙ-Driven Drug Repurposing: Applications and Challenges [https://pmc.ncbi.nlm.nih.gov/articles/PMC12641654/]
- 50. repositioning: Progress and challenges in Drug for... drug discovery [https://pmc.ncbi.nlm.nih.gov/articles/PMC8883737/]
- 51. DTIAM: a unified framework for predicting drug-target ... [https://www.nature.com/articles/s41467-025-57828-0]
- 52. Repurposing FDA approved drugs for psoriasis indications ... [https://www.nature.com/articles/s41598-025-01448-7]
- 53. Exploring drug-target interaction prediction on cold-start ... [https://www.sciencedirect.com/science/article/abs/pii/S1046202324002470]
- 54. A unified drug–target interaction prediction framework ... [https://www.nature.com/articles/s41467-021-27137-3]
- 55. DRUG-TARGET INTERACTION/AFFINITY PREDICTION [https://arxiv.org/html/2502.15346v1]
- 56. An Industrial Perspective on Ligand Bias in GPCR Drug ... [https://www.sciencedirect.com/science/article/abs/pii/S0028390825004903]
- 57. Structure-Based vs Ligand-Based Drug Design [https://www.iaanalysis.com/structure-based-vs-ligand-based-drug-design.html]
- 58. Merging Ligand-Based and Structure-Based Methods in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7587536/]
- 59. Biased ligand quantification in drug discovery [https://pmc.ncbi.nlm.nih.gov/articles/PMC4940816/]
- 60. Ligand-Induced Biased Activation of GPCRs [https://pubmed.ncbi.nlm.nih.gov/40076272/]
- 61. 3D molecular generative framework for interaction-guided ... [https://www.nature.com/articles/s41467-024-47011-2]
- 62. Latent Biases in Machine Learning Models for Predicting ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9850481/]
- 63. Highly biased agonism for GPCR ligands via nanobody ... [https://www.nature.com/articles/s41467-024-49068-5]
- 64. Drug-target interaction prediction using ensemble learning ... [https://www.sciencedirect.com/science/article/abs/pii/S1046202317300257]
- 65. Drug-Protein Interactions Prediction Models Using Feature ... Selection [https://pubmed.ncbi.nlm.nih.gov/38270152/]
- 66. In-silico target prediction by ensemble chemogenomic model ... [https://jcheminf.biomedcentral.com/articles/10.1186/s13321-023-00720-0]
- 67. Hybrid computational–experimental data-driven design of self ... [https://pubs.rsc.org/en/content/articlehtml/2022/dd/d1dd00047k]
- 68. DTA-GTOmega: Enhancing Drug-Target Binding Affinity ... [https://www.sciencedirect.com/science/article/abs/pii/S0022283624004728]
- 69. Drug-target binding affinity prediction based on power graph ... [https://bmcmedgenomics.biomedcentral.com/articles/10.1186/s12920-024-02073-5]
- 70. Benchmarking compound activity prediction for real-world ... [https://www.nature.com/articles/s42004-024-01204-4]
- 71. MolScore: a scoring, evaluation and benchmarking framework ... [https://jcheminf.biomedcentral.com/articles/10.1186/s13321-024-00861-w]
- 72. Stacked variational autoencoder with SMILES-based drug ... [https://www.sciencedirect.com/science/article/abs/pii/S0925231225025093]
- 73. Enhancing the confidence of potential targets enriched by ... [https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2025.1574540/full]
- 74. Comprehensive detection and characterization of human ... [https://www.nature.com/articles/s41467-024-52146-3]
- 75. Accurate prediction of protein–ligand interactions by ... [https://jcheminf.biomedcentral.com/articles/10.1186/s13321-024-00912-2]
- 76. Drug-target interaction prediction using reliable negative ... [https://www.sciencedirect.com/science/article/abs/pii/S1056871922000387]
- 77. Challenges, Applications, and Recent Advances of Protein ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6270832/]
- 78. Merging Ligand-Based and Structure-Based Methods in ... [https://www.mdpi.com/1420-3049/25/20/4723]
- 79. How to approach machine learning-based prediction of drug ... [https://jcheminf.biomedcentral.com/articles/10.1186/s13321-023-00689-w]
- 80. Machine Learning for Multi-Target Drug Discovery [https://pmc.ncbi.nlm.nih.gov/articles/PMC12473769/]
- 81. Computational approaches streamlining drug discovery [https://www.nature.com/articles/s41586-023-05905-z]
- 82. Structure-based discovery and experimental validation of ... [https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2025.1605741/full]
- 83. DeepDTAGen: a multitask deep learning framework for ... [https://www.nature.com/articles/s41467-025-59917-6]
- 84. Top 5 Drug Discovery Trends 2025 Driving Breakthroughs [https://www.pelagobio.com/cetsa-drug-discovery-resources/blog/drug-discovery-trends-2025/]
- 85. Inside ELRIG's Drug Discovery 2025 [https://www.drugtargetreview.com/article/190237/inside-elrigs-drug-discovery-2025/]